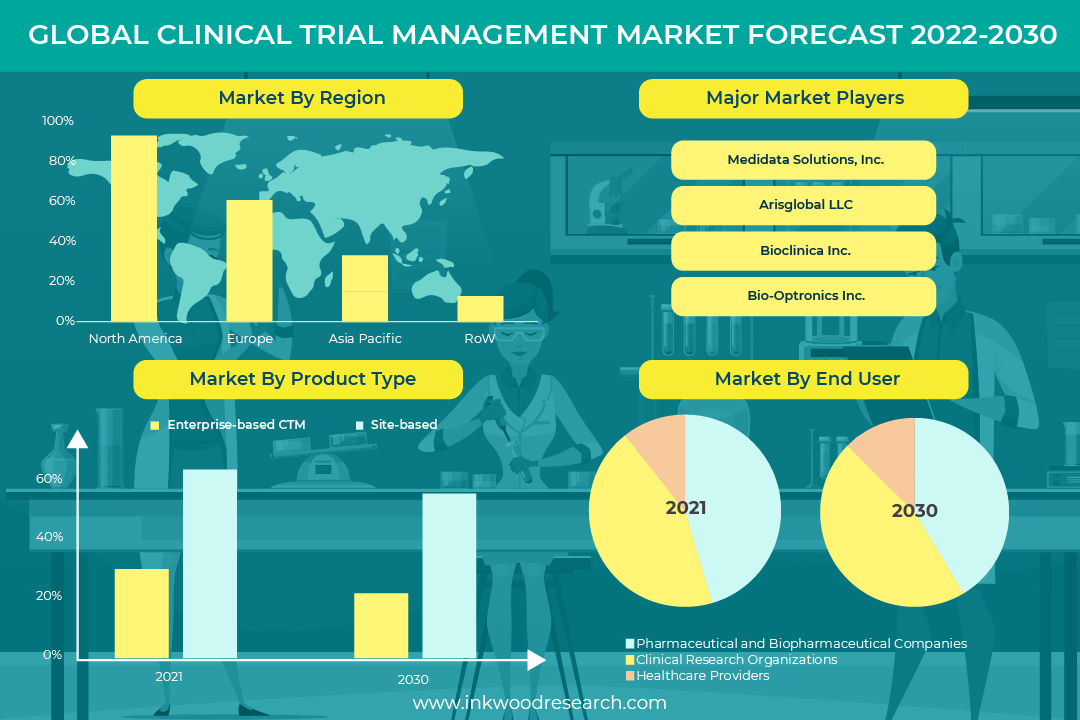
According to a new market research report launched by Inkwood Research, the Global Clinical Trial Management Market is set to reach around $2623.27 million by 2030, growing at a CAGR of 12.71% over the forecasting years.
Browse 48 Market Data Tables and 46 Figures spread over 160 Pages, along with in-depth analysis on Global Clinical Trial Management Market by Product Type, Deployment Model, Component, End-User, & by Geography
This insightful market research report by Inkwood Research focuses on market trends, leading players, supply chain trends, technological innovations, key developments, and future strategies. The report covers all the aspects of this comprehensive market by assessing major geographies and is a valuable asset for the existing players, new entrants, and future investors. The study presents a detailed market analysis, with inputs derived from industry professionals across the value chain.
Global Clinical Trial Management Market Scenario
A clinical trial management system (CTMS) is a software system used by pharmaceutical and biotechnology industries to manage their clinical trials. Also, it can store enterprise information, act as a central repository for the organization’s key information, and manage several clinical, operational processes.
Several teams and researchers engaged in clinical trials benefit from CTMS with regard to the maintenance and management of clinical trials. CTMS tracks and manages activities in a clinical trial, offering a real-time preview of the different operations. It also enables a level of transparency between finance and study teams, thereby reducing the costs associated with managing clinical trials. Such aspects also impact the accuracy and efficiency of the managing process.
Further, CTMS helps manage time effectively for clinical trial processes. Also, it acts as a substitute for paper-based and skillful processes in clinical trials. The other benefits of CTMS include enhanced productivity and work quality. Thus, the growing awareness of CTMS’ benefits among pharmaceutical companies and research institutes drives the market growth. However, security concerns and implementations costs hamper the market growth.
The Global Clinical Trial Management Market report provides data tables and includes charts and graphs for visual analysis.
Request Free Sample Report for Global Clinical Trial Management Market by Product Type, Deployment Model, Component, End-User, & by Geography: https://inkwoodresearch.com/reports/clinical-trial-management-market/#request-free-sample
Market Segmentation
Market by End-User
- Healthcare Providers
- Pharmaceutical and Biopharmaceutical Companies
- Clinical Research Organizations
Market by Product Type
- Site-Based CTM
- Enterprise-Based CTM
Market by Deployment Model
- Cloud-Based Deployment
- On-Premise Deployment
- Web-Based Deployment
Market by Component
- Services
- Hardware
- Software
Report Highlights
– The report provides a detailed analysis of the current and future market trends to identify the investment opportunities – Market forecasts till 2028, using estimated market values as the base numbers – Key market trends across the business segments, regions, and countries – Key developments and strategies observed in the market – Market dynamics such as drivers, restraints, opportunities, and other trends – In-depth company profiles of key players and upcoming prominent players – Growth prospects among the emerging nations through 2028
Companies Profiled
- MEDIDATA SOLUTIONS INC
- VEEVA SYSTEMS INC
- BIO-OPTRONICS INC
- MEDNET SOLUTIONS INC
- BIOCLINICA INC
- ORACLE CORPORATION
- IBM CORPORATION
- ADVARRA (FORTE RESEARCH SYSTEMS INC)
- ARISGLOBAL LLC
- PAREXEL INTERNATIONAL CORPORATION
Related Reports
GLOBAL LABORATORY AUTOMATION MARKET – The global laboratory automation market is projected to register a CAGR of 5.61% during the forecast period, 2021-2028.
Media Contact
Company Name: Inkwood Research
Contact Person: Arvind Iyer
Email: Send Email
Phone: 1-(857) 293-0150
Country: United States
Website: https://www.inkwoodresearch.com/