Roots Analysis has announced the addition of “Lyophilization Services Market for Biopharmaceuticals (2nd Edition), 2022 – 2035” report to its list of offerings.
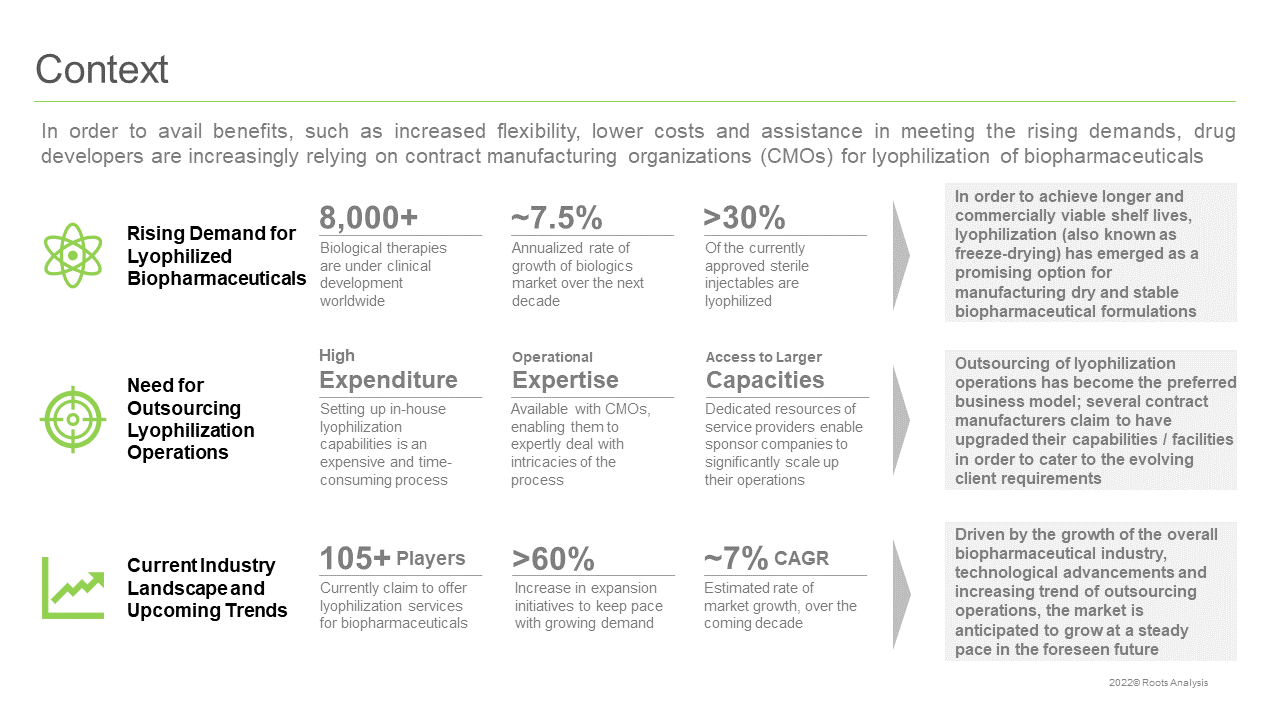
In order to achieve longer and commercially viable shelf lives, lyophilization has emerged as the preferred approach, enabling the manufacturing of stable and dry biopharmaceutical formulations. Presently, a number of biopharmaceutical companies are outsourcing their lyophilization operations to contract service providers that have dedicated facilities, equipment and production lines for this purpose. Further, the technological advancements in this domain have resulted in improved productivity and efficiency of the overall process.
To order this 220+ page report, which features 80+ figures and 140+ tables, please visit
Key Market Insights
More than 105 companies claim to offer lyophilization services for biopharmaceuticals, globally
Majority (48%) of the industry stakeholders are based in North America, followed by those having headquarters in Europe (42%). In addition, more than 40% of the service providers have the required capabilities to carry out the lyophilization of proteins, followed by companies offering lyophilization services for antibodies (32%) and vaccines (21%).
More than 55% of partnership agreements were inked in 2020 and 2021
Majority of the agreements related to lyophilization of biopharmaceuticals were observed to be instances of acquisitions, representing over 60% of the total partnerships. Further, most of the intercontinental, as well as intracontinental, deals have been inked by players based in North America.
Expansion activity in this domain has grown at a CAGR of ~28%, between 2018 and 2021
Around 55% of the initiatives were focused on addition of lyophilization capacity, followed by instances of facility expansions (32%) and new facility additions (14%). More than 85% of the expansions reported in this domain were local initiatives, followed by international initiatives (12%).
North America and Europe are anticipated to capture more than 70% of the market share, by 2035
In addition, the market in Asia Pacific is likely to grow at a relatively faster pace (7.8%), in the long term. Further, in 2035, majority share (52%) of the overall biopharmaceutical lyophilization market is likely to be captured by antibodies.
To request a sample copy / brochure of this report, please visit
https://www.rootsanalysis.com/reports/161/request-sample.html
Key Questions Answered
- Who are the key players engaged in offering lyophilization services for biopharmaceuticals?
- What is the relative competitiveness of lyophilization service providers for biopharmaceuticals?
- What types of partnership models are commonly adopted by stakeholders in this industry?
- What are the different types of expansion initiatives being undertaken by lyophilization service providers for biopharmaceuticals?
- What are the key market trends and driving factors that are likely to impact this market?
- How is the current and future market opportunity likely to be distributed across key segments?
The financial opportunity within the lyophilization services market for biopharmaceuticals has been analyzed across the following segments:
Type of Biologic Lyophilized
- Antibodies
- Cell and Gene Therapies
- Vaccines
- Other Bioloigics
Type of Primary Packaging System
- Vials
- Syringes
- Cartridges
- Ampoules
- Others
Key Geographical Regions
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and North Africa
- Rest of the World
The report also features inputs from eminent industry stakeholders, according to whom, continuous growth of the biological therapeutics pipeline and increasing preference for outsourcing is likely to drive the growth of the overall lyophilization service providers market, in the coming future. The report includes detailed transcripts of discussions held with the following experts:
- Brian Sasaki (Director of Business Development) and Thomas Ingallinera (Vice President of Technical Support, Pharmaceutical International)
- Doreen Coull (Regional Sales Director, Curia)
- Patrick DePalma (Former Director of CMO Business Development, BioDefense Division, Emergent BioSolutions)
- Rocio Gonzalez Rodriguez (Head of CDMO Project Management, Liof Pharma)
- John Shaw (Former Head of Business Development and Marketing, Vibalogics)
- Sophia Kim (Commercial DP (Depository Participant) Sales / Group Leader, Samsung Biologics)
- Marianna Tcherpakov (Business Development Manager, Bioserv)
- Adam Inche (Marketing Manager, Symbiosis Pharmaceutical Services)
- Adam Viverette (Lyophilization Manager, Patheon)
- Steven Nail (Principal Scientist, Baxter BioPharma Solutions)
The research includes profiles of key players (listed below); each profile features a brief overview of the company, its financial performance (if available), recent developments and an informed future outlook.
- Baxter BioPharma Solutions
- BioZed Engineering
- CinnaGen
- CordenPharma
- Coriolis Pharma
- Curia
- Emergent BioSolutions
- LSNE Contract Manufacturing
- Lyophilization Technology
- MabPlex
- Northway Biotech
- ProJect Pharmaceutics
- Samsung Biologics
- Vetter Pharma
- WuXi Biologics
For additional details, please visit
https://www.rootsanalysis.com/reports/view_document/lyophilization-services-market-for-biopharmaceuticals/161.html or email sales@rootsanalysis.com
You may also be interested in the following titles:
- Pharmaceutical Secondary Packaging Market: Industry Trends and Global Forecasts, 2022-2035
- Continuous Manufacturing (Small Molecules and Biologics) Market (2nd Edition): Industry Trends and Global Forecasts, 2022-2035
- Live Biotherapeutic Products and Microbiome Manufacturing Market: Industry Trends and Global Forecasts, 2022-2035
- Peptide Therapeutics: Contract API Manufacturing Market: Industry Trends and Global Forecasts, 2022-2035
- Biopharmaceutical Excipient Manufacturing Market: Industry Trends and Global Forecasts, 2022-2035
- Companion Diagnostics Development Services Market: Industry Trends and Global Forecasts, 2022-2035
Media Contact
Company Name: Roots Analysis
Contact Person: Gaurav Chaudhary
Email: Send Email
Phone: +1 (415) 800 3415, +44 (122) 391 1091
City: London
Country: United Kingdom
Website: www.rootsanalysis.com