MEDICA 2022 is taking place from Nov 14 to 17 in Dusseldorf Germany. It is a comprehensive medical exhibition recognized as the leading event in the medical sector worldwide, drawing 4200 companies from 70 countries exhibiting at the show.
CreativeBio is showcasing the following brands at the expo: 1) Colosafe®, a stool DNA test kit for early detection of colorectal cancer; 2) Urisafe, a noninvasive DNA test kit for early detection of bladder cancer; 3) Lunsafe, a noninvasive DNA test kit for early detection of lung cancer; 4) Automatic Sample Preprocessing Instrument; 5)HelixPOC® Automated Microfluidic qPCR System; 6) HemoPOC®, a POCT device for self monitor anti-coagulation.
CreativeBio would like to extend their invitation to everyone from all corners of the world to visit them at booth # 17D46-4.

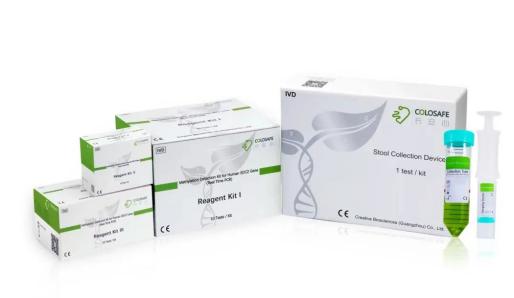
Colosafe®, methylation detection kit for human SDC2 gene and stool collection device, was cleared by National Medical Products Administration on Nov 20, 2018. Thus it is the first of its kind approved by NMPA and the ice-breaking noninvasive sDNA test for early detection of colorectal cancer in China. It is a product that can precisely capture and interpret the mutated messages (human SDC2 methylation) from stool to help physicians to detect the cancerous lesion early and prevent the development of colorectal cancer at early stage.
HelixPOC Automated Microfluidic qPCR System combines microfluidics with real time polymerase chain reaction technology. A reagent preloaded microfluidic cartridge controlled by an analyzer allows the entire nuclear acid detection process to be accomplished fully automatically in an enclosed system, including sample preprocessing, DNA extraction, PCR amplification, fluorescence measurement and data analysis. Such a fully automated and fully enclosed point-of-care (POCT) system makes it possible to conduct molecular testing anywhere in a sample-in-result-out manner.

HemoPOC® is developed by Hilixgen, a subsidiary company wholly owned by CreativBio. This coagulation analyzer, coupled with disposable PT-INR test strips, yields immediate results for the quantitative testing of prothrombin time, allowing patients undergoing oral anti-coagulation therapy of Warfarin to monitor their International Normalized Ratio(INR) using a finger-prick blood test at the point of care. This handheld device can be used at clinic as well as at home to monitor and self monitor anti-coagulation anytime and anywhere. It has CE Mark for international market where CE is accepted.
Media Contact
Company Name: Creative Biosciences (Guangzhou) CO., Ltd
Contact Person: Liu Yan
Email: Send Email
Phone: +86-020-82037860
Address:2nd Floor, Buildiing A2, No. 11 Kaiyuan Avenue, Science City Guangzhou High-Tech Industrial Development Zone
City: Guangzhou
State: Guangdong, 510530
Country: China
Website: https://en.creativebio.cn/