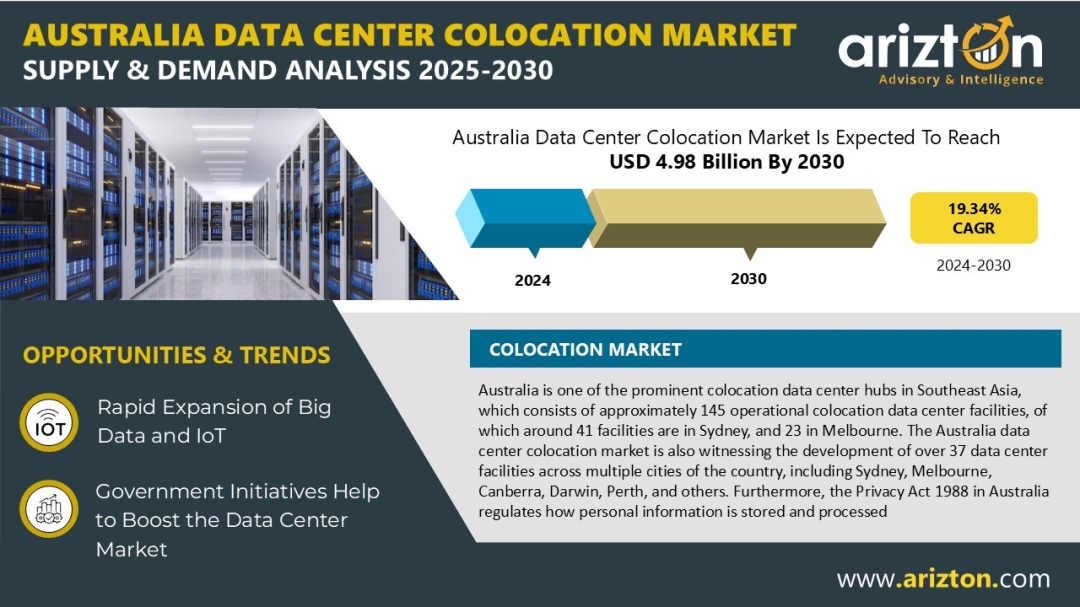
According to Arizton latest research report, the Australia data center colocation market to grow at a CAGR of 19.34% during the forecast period.
Looking for More Information? Click: https://www.arizton.com/market-reports/australia-data-center-colocation-market
Report Scope:
MARKET SIZE – COLOCATION REVENUE: USD 4.98 billion (2030)
CAGR – COLOCATION REVENUE:19.34% (2024-2030)
MARKET SIZE – UTILIZED WHITE FLOOR AREA: 15.76 million sq. feet (2030)
MARKET SIZE – UTILIZED RACKS: 309.55 thousand units (2030)
MARKET SIZE – UTILIZED IT POWER CAPACITY: 3,480 MW (2030)
BASE YEAR: 2024
FORECAST YEAR: 2025-2030
Australia Digital Economy Accelerates with AI-Ready Data Center Investments
Australia is entering a transformative phase as AI adoption and large-scale digitalization reshape enterprise operations and consumer behavior. The emergence of AI factories, advanced energy management platforms, and a $1.7 billion federal commitment to digital innovation is catalyzing demand for next-generation data centers across the country. These facilities are being designed with liquid cooling systems, renewable energy integration, and AI-optimized infrastructure to handle the exponential growth in high-performance computing and large language model (LLM) workloads.
With global cloud and colocation providers expanding their AI infrastructure footprints in Melbourne, Sydney, and Perth, Australia is fast consolidating its position as a strategic APAC hub for AI-ready and sustainable digital infrastructure.
Recent Developments in Australia Data Center Market:
-
In April 2025, STACK Infrastructure announced plans to develop a new data center facility in Sydney, offering approximately 450 MW of power capacity.
-
In the same month, Goodman revealed plans for a new data center campus in Sydney with a combined IT capacity of 90 MW.
-
In October 2024, NEXTDC announced its S7 data center campus in Sydney, expected to deliver 550 MW of power capacity.
-
In August 2024, NEXTDC launched a new facility in Darwin, Northern Australia, providing 7 MW of power capacity.
Australia Data Centers Redefine Growth Amid Energy Strain and Net-Zero Goals
Australia is fast-tracking its data center colocation market into a greener future as the nation pursues its ambitious net-zero 2050 target. By 2025, renewable sources are expected to supply 48% of the country’s electricity, rising to 82% by 2030, creating a strong foundation for sustainable digital infrastructure. With this shift, the industry faces growing challenges, including high power consumption and operational risks from Australia’s exposure to natural disasters such as floods, storms, and cyclones, which can disrupt uptime.
To address these concerns, the Australian government has strengthened regulations, mandating a five-star NABERS rating for data centers from mid-2025 and launching a new Data Centre Panel to promote sustainable practices and reduce emissions across the sector. In response, Operators are responding with long-term renewable energy agreements and investments in resilient infrastructure, transforming Australia into a sustainable, future-ready hub for digital infrastructure in APAC and attracting hyperscalers, cloud providers, and enterprises seeking reliable, green operations.
Download the full report for strategic insights, segment forecasts, and growth opportunities:https://www.arizton.com/market-reports/australia-data-center-colocation-market
Vendor Landscape
Existing Colocation Operators
-
AirTrunk
-
NEXTDC
-
CDC Data Centres
-
STACK Infrastructure
-
Macquarie Data Centres
-
DCI DATA CENTERS
-
Equinix
-
Global Switch
-
Fujitsu
-
Digital Realty
-
Telstra InfraCo
-
Keppel DC REIT
-
Polaris Data Centre
-
Others
New Operators
-
Quinbrook Infrastructure Partners
-
Goodman
-
GreenSquareDC
-
ISPT
-
Cundall
-
LCI Consultants
-
DOMA Infrastructure Group
-
Vantage Data Centers
-
MARKHAM
-
Trifalga
Other Related Reports that Might be of Your Business Requirement
Vietnam Data Center Colocation Market – Supply & Demand Analysis 2025-2030
https://www.arizton.com/market-reports/vietnam-data-center-colocation-market
New Zealand Data Center Colocation Market – Supply and Demand Analysis 2025-2030
https://www.arizton.com/market-reports/new-zealand-data-center-colocation-market
Key Questions Answered in the Report:
-
What factors are driving the Australia data center colocation market?
-
How much MW of IT power capacity is likely to be utilized in Australia by 2030?
-
What is the count of existing and upcoming colocation data center facilities in Australia?
-
Who are the new entrants in the Australia data center industry?
-
What is the count of existing and upcoming colocation data center facilities in Australia?
-
How much MW of IT power capacity is likely to be utilized in Australia by 2030?
-
What factors are driving the Australia data center colocation market?
-
Who are the new entrants in the Australia data center industry?
What’s Included in the Australia Data Center Colocation Market Report?
The report offers transparent research on Australia’s data center colocation market, covering market size by white floor area, IT power capacity, racks, and occupancy. It analyzes current and future colocation demand across industries, cloud operations, and sustainability, with insights on 145 existing and 37 upcoming facilities across 21+ cities. The study includes market revenue and forecasts (2024–2030) for retail and wholesale colocation, pricing trends, key growth opportunities, challenges, competitive and vendor landscapes, and a snapshot of submarine cables and cloud-on-ramp services supporting the ecosystem.
Why Arizton?
100% Customer Satisfaction
24×7 availability – we are always there when you need us
200+ Fortune 500 Companies trust Arizton’s report
80% of our reports are exclusive and first in the industry
100% more data and analysis
1500+ reports published till date
Post-Purchase Benefit
-
1hr of free analyst discussion
-
10% off on customization
About Us:
Arizton Advisory and Intelligence is an innovative and quality-driven firm that offers cutting-edge research solutions to clients worldwide. We excel in providing comprehensive market intelligence reports and advisory and consulting services.
We offer comprehensive market research reports on consumer goods & retail technology, automotive and mobility, smart tech, healthcare, life sciences, industrial machinery, chemicals, materials, I.T. and media, logistics, and packaging. These reports contain detailed industry analysis, market size, share, growth drivers, and trend forecasts.
Arizton comprises a team of exuberant and well-experienced analysts who have mastered generating incisive reports. Our specialist analysts possess exemplary skills in market research. We train our team in advanced research practices, techniques, and ethics to outperform in fabricating impregnable research reports.
Media Contact
Company Name: Arizton Advisory & Intelligence
Contact Person: Jessica
Email: Send Email
Phone: +1 3122332770
Country: United States
Website: https://www.arizton.com/market-reports/australia-data-center-colocation-market
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Australia Data Center Colocation Market to Exceed USD 4.98 Billion by 2030 Amid AI and Sustainable Infrastructure Expansion