Dymesty today unveiled its AI Glasses on Kickstarter, introducing the world’s first titanium smart eyewear designed to enhance productivity for business professionals through hands-free AI assistance, meeting recording, and real-time translation capabilities.

Dymesty AI Glasses are ultra-lightweight smart eyewear that seamlessly integrate AI-powered productivity tools into a 35g titanium frame, enabling business professionals to capture meetings, translate conversations in real-time, and manage communications hands-free while maintaining professional style and all-day comfort.
In today’s hyper-connected business environment, professionals lose critical insights while switching between devices, missing 40% of meeting content during note-taking, and struggling with language barriers in global teams. Traditional smart glasses fail to address these challenges—they’re too heavy (50-70g average), require frequent charging (4-6 hours), and have a plastic feel, looking cheap and unprofessional.
Dymesty AI Glasses solve these productivity gaps through three breakthrough innovations:
1. Ultra-light 35g design eliminates fatigue during 10+ hour workdays
2. 48-hour battery life covers entire business trips without charging anxiety
3. Invisible AI integration maintains a professional appearance in any setting
“When we designed the Dymesty AI Glasses, we wanted something really useful for everyday life—like picking up calls, getting instant translations, and staying connected without always looking at your phone. Our goal was simple: create something natural, stylish, and that actually makes your day easier. Something you’d want to wear on a daily basis,” the Dymesty team explains, reflecting on how they built the world’s first titanium AI glasses. “When we talk about AI glasses, most people think bulky, awkward, or overhyped. For a business professional who needs to maintain a polished image, what we need, first and foremost, is a good pair of glasses that are comfortable to wear.”

How Dymesty AI Glasses Transform Your Workday
Intuitive Gesture Controls:
1. Double-tap temple: Double tap to wake your AI Assistant.
2. One-second hold: Start meeting recording with AI transcription
3. Three-second hold: Activate private audio mode for confidential calls
Real-World Productivity Scenarios:
1. Client meetings: Auto-record, transcribe, and generate a clear summary
2. International communications: Real-time translation across 100+ languages
3. Evening flights: 48-hour battery means no charging between destinations
Core Specifications
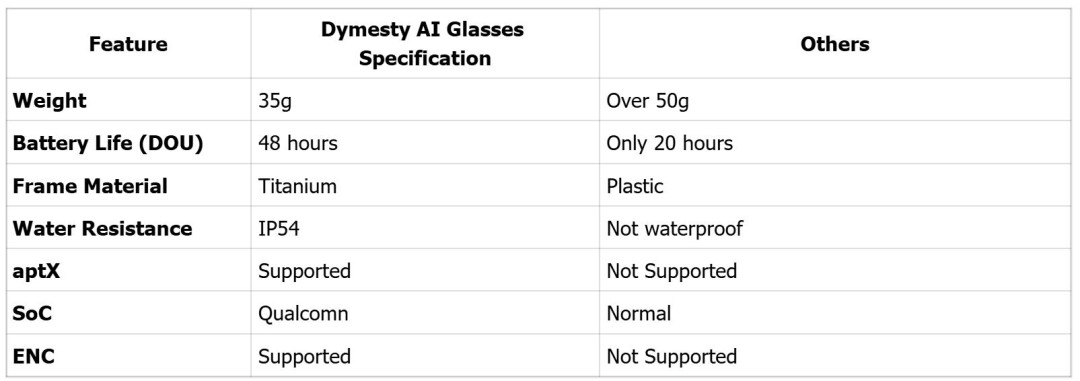
Key Features in Detail
Titanium Construction: The titanium frame with hypoallergenic silicone nose pads ensures all-day comfort. Featuring a proprietary U-shaped titanium alloy spring hinge and an ergonomic hug-style temple design, Dymesty is designed to fit a variety of face shapes.
Smart Recording & Transcription: Capture meetings, lectures, and conversations with only a long press for one second. The companion app automatically transcribes audio & creates searchable notes, and syncs across your computer through the app.
Real-Time Translation: Supporting 100+ languages, including English, Mandarin, Spanish, French, German, and Japanese etc. Translation accuracy verified at 95% by independent testing.
Premium Audio Experience: Environmental Noise Cancellation (ENC) combined with aptX HD delivers crystal-clear calls even in airports or cafes. Directional speakers ensure privacy while maintaining awareness of surroundings.
48-Hour Intelligent Battery: Advanced power management achieves 48 hours of typical use (8 hours meeting time or 12 hours of continuous music).
Frequently Asked Questions (FAQ)
Q1: How comfortable are Dymesty AI Glasses for all-day wear?
A: At just 35g, they’re lighter than most AI glasses (over 60g). With a U-shaped titanium alloy spring hinge and an ergonomic hug-style temple design, Dymesty is designed to fit a variety of face shapes. With the help of this flexible, precisely balanced structure, your glasses will stay in place all day without slipping or creating pressure points.
Q2: Can I add prescription lenses to Dymesty AI Glasses?
A: Absolutely! The frame accepts standard prescription lenses at any optical store. We also offer a prescription lens service starting at $99 for single vision and $179 for progressive lenses(all include Ultra-thin 1.74 Index Lens).
Q3: How does meeting recording work? Is it legal?
A: Long press for 1s to start recording. The AI automatically transcribes speech, identifies speakers, and generates summaries. All recordings are encrypted end-to-end. Regarding legality: always follow local recording consent laws. The glasses include consent notifications.
Q4: Which languages does real-time translation support?
A: Currently 100+ languages including English, Mandarin, Spanish, French, German, Japanese, Korean, Arabic, Hindi, Portuguese, Russian, and Italian etc. Translation accuracy is over 95% for daily use.
Q5: How do Dymesty glasses compare to others?
A: Key differences:
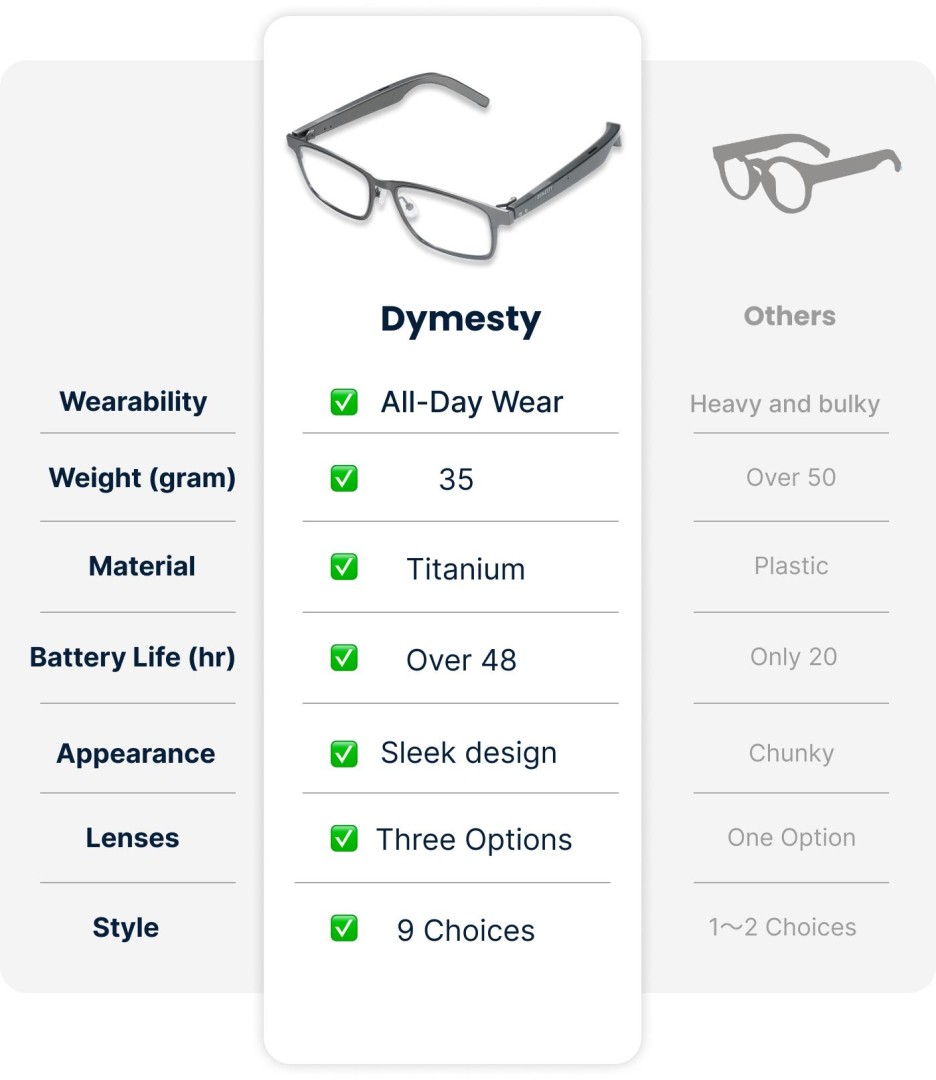
Q6: What’s the delivery timeline for Kickstarter backers?
A: Manufacturing timeline:
1. September 2024: Making the first sample
2. July 2025: Production start
3. October 2025: Super Early Bird shipments
4. November 2025: All backer shipments
Q7: What happens if the Kickstarter campaign doesn’t reach its goal?
A: Kickstarter uses all-or-nothing funding, so you’re only charged if Dymesty reaches its $10,000 goal. All backers are protected by Kickstarter’s policies.
Kickstarter Campaign Details
Campaign Timeline
Launch: August 19, 2025, 10:00AM EST
Duration: 45 days
Reward Tiers
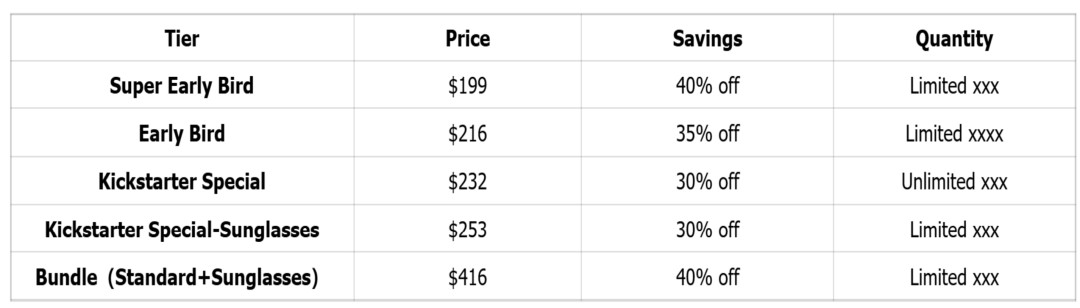
Stretch Goals
1. $10K: Base goal – Production confirmed
2. $200K: Premium case for all backers
3. $500K: Additional color – Gray & Golden
4. $1M: Additional color – Black & Blue
5. $1.5M: Additional color – Twilight
6. $2M: Additional color – Titanium Gray & Golden
Dymesty glasses are being manufactured in high-quality, well-established factories with reliable production capacity and are undergoing thorough preparations for mass production. After rigorous research and testing phases, Dymesty is now working hard to ensure a smooth production process and dependable delivery to our global backers.
About Dymesty
Dymesty redefines AI glasses. Designed for everyday comfort, discreet intelligence, and timeless business elegance, Dymesty transcends wearable tech — delivering a premium eyewear experience powered by AI. No more awkward gadgets.
We created Dymesty AI glasses professionals genuinely desire to wear daily. Proudly introducing exquisitely crafted frames with sophisticated always-on functionality. Rejecting clunky plastics and poor ergonomics, our team fused premium eyewear design with cutting-edge AI — resulting in an experience as beautiful as it is intelligent.
Inspired by technology’s relentless tide, the Dymesty team empowers those striving for excellence, embracing life, and daring to dream. Our mission is to help people stay ahead of innovation and delight in its possibilities. With real-time transcription, translation, navigation, smart tasks, and phone-left alerts, Dymesty opens a new chapter in personalized, intelligent living.
Born from a vision of a smarter, more convenient, and more beautiful world, we integrate AI to infuse joy, efficiency, and inspiration into every moment — redefining tech-enabled lifestyles.
For the latest news and updates from Dymesty, please follow the official Dymesty channels on social media:
Facebook: https://www.facebook.com/profile.php?id=100089380524675
YouTube: https://www.youtube.com/@Dymesty
Instagram:https://www.instagram.com/dymesty/
Media Contact
Company Name: Dymesty
Contact Person: Crystal Lin
Email: Send Email
Country: United States
Website: https://dymesty.com/