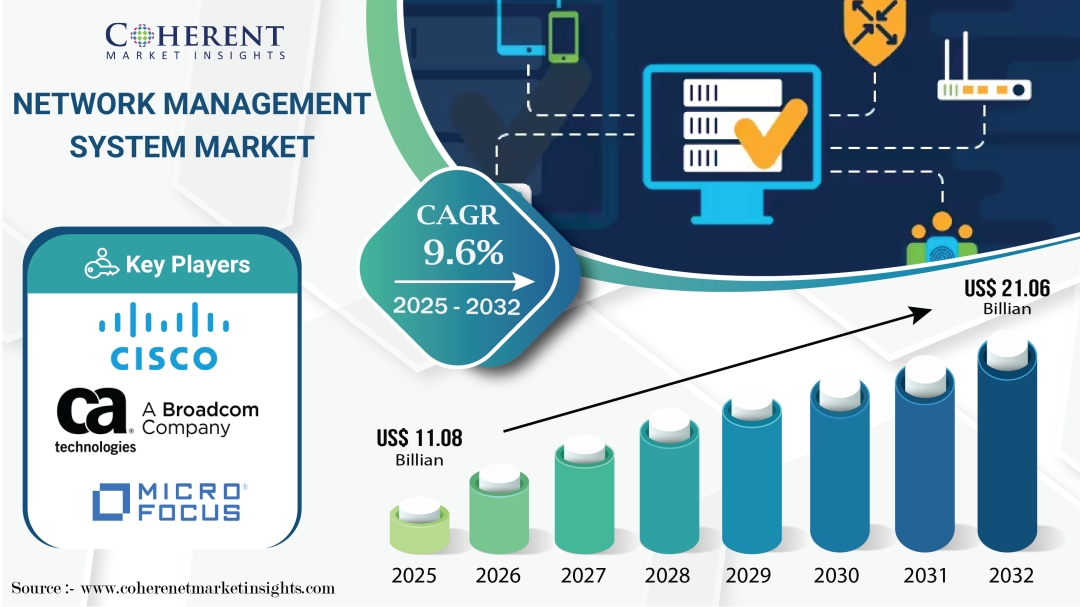
Network Management System Market is estimated to be valued at USD 11.08 Bn in 2025 and is expected to reach USD 21.06 Bn in 2032, exhibiting a compound annual growth rate (CAGR) of 9.6% from 2025 to 2032. The growing adoption of automation technologies across diverse industries, combined with increasing network complexity, is driving demand for advanced network management systems. Between 2022 and 2025, significant advancements in artificial intelligence (AI) and machine learning have enhanced network analytics and automation, leading to improved operational efficiency. Additionally, the rapid proliferation of IoT devices, ongoing 5G deployment, and heightened demand for robust cybersecurity solutions are expected to further propel market growth.
Request Sample Pages: https://www.coherentmarketinsights.com/insight/request-sample/7511
Global Network Management System Market Key Takeaways
According to Coherent Market Insights (CMI), the global network management system market size is expected to grow at a CAGR of 9.6%, increasing from USD 11.08 Bn in 2025 to USD 21.06 Bn by 2032.
By component, solution segment is expected to dominate the industry, accounting for a market share of 68.4% in 2025.
Based on deployment, on-premises category is likely to hold a prominent market share of 56.2% in 2025.
Large enterprises segment is projected to account for more than half of the global network management system market share in 2025.
North America is set to lead the global market, accounting for over one-third of the industry share in 2025.
As per CMI’s new network management system market analysis, Asia Pacific is expected to experience fastest growth during the projection period.
Rising Network Complexity Fueling Market Growth
Coherent Market Insights’ latest network management system market research report offers insights into major factors driving industry growth. Increasing network complexity is one such key growth factor.
Rising penetration of connected devices is making modern networks more complex. This is expected to drive demand for network management systems, as these solutions enable businesses to monitor, manage, and optimize their increasingly complicated network infrastructures.
High Costs Limiting Network Management System Market Growth
The future network management system market outlook looks promising. However, growing high initial investment costs could somewhat constrain market expansion over the forecast period.
Advanced NMS solutions require significant upfront expenditures in hardware and software components. As a result, many organizations, particularly small and medium-sized enterprises, struggle to allocate the necessary budget for implementation. This financial barrier could slightly slow down network management system market demand during the forecast period.
Increasing Cybersecurity Threats Unlocking New Growth Opportunities
Cyberattacks are becoming more sophisticated, with modern cybercriminals employing advanced technologies like AI to create deepfakes and automated attack strategies. As a result, ensuring robust network security remains a top priority for businesses.
Network management system (NMS) solutions play a key role in enhancing cybersecurity. They help to identify vulnerabilities, monitor unusual network activities, and ensure compliance with security protocols.
Some NMS platforms incorporate advanced features like intrusion detection and prevention systems (IDPS), further strengthening network defenses. Thus, rising incidence of cyberattacks is expected to create lucrative growth opportunities for network management system companies during the forecast period.
Get Customization on this Report: https://www.coherentmarketinsights.com/insight/request-customization/7511
Impact of AI on the Network Management System Market
Artificial intelligence (AI) is becoming a powerful tool in the network management system (NMS) market. This advanced technology drives automation, enhances predictive capabilities, and bolsters security.
AI-enabled tools help automate routine tasks like device configuration, traffic routing, and load balancing. By doing so, they significantly reduce operational overhead and minimize human error.
Similarly, predictive analytics powered by machine learning can forecast network issues before they occur. This enables pre-emptive maintenance and reduce downtime.
Real-time monitoring and anomaly detection tools swiftly identify and resolve performance dips or security threats. They help businesses optimize bandwidth usage and improve user experience. When it comes to security, AI-driven NMS significantly enhances threat detection and response times.
Emerging Network Management System Market Trends
Rising popularity of cloud-based network management systems is a key trend in the market. Cloud-based NMS solutions are gaining immense traction due to the broader shift toward cloud computing, offering advantages like scalability, ease of deployment, and cost-effectiveness.
Increasing penetration of 5G, especially across nations like China, is expected to boost growth of the network management system market. 5G’s need for low latency and high bandwidth demands advanced NMS tools to manage complex networks, real-time traffic, and key applications like IoT, self-driving tech, and AR/VR.
Emergence of software-defined networking (SDN) and network function virtualization (NFV) presents significant opportunities for network management system vendors. Enterprises are increasingly adopting SDN and NFV to modernize their networks, enhance agility, reduce operational costs, and accelerate service innovation. In response, NMS vendors are integrating advanced capabilities for programmability, abstraction, and orchestration to manage virtualized infrastructure and network functions effectively.
Ongoing digital transformation initiatives will likely play a key role in boosting the network management system market value. As organizations undergo rapid digitalization, they rely more on robust, scalable, real-time networks. This is driving demand for advanced NMS solutions to ensure connectivity, security, and performance.
Analyst’s View
“The global network management system industry is set for robust growth, owing to increasing network complexity, rising incidence of cyberattacks, growing popularity of cloud-based NMS solutions, and expanding adoption of IoT and connected devices across industries,” said Ankur Rai, a senior analyst at CMI.
Current Events and Their Impact on the Network Management System Market
|
Event |
Description and Impact |
|
Commercialization of 5.5G and AI-Native Networks |
|
|
Rising State-Sponsored Cyberattacks |
|
|
AI Integration into Network Operations (AIOps) |
|
Competitor Insights
Key companies in the network management system market report:
– Broadcom CA Technologies
– Cisco
– Paessler
– Micro Focus
– Nokia
– Juniper Networks
– Progress
– Huawei
– Kentik
– HelpSystems
– Ericsson
– AppNeta
– Dell Technologies, Inc.
– Hewlett Packard Enterprise Company
Buy this Complete Business Research Report: https://www.coherentmarketinsights.com/insight/buy-now/7511
Key Developments
In June 2025, Cisco introduced a new secure network architecture aimed at accelerating workplace AI transformation. The new network architecture simplifies network operations, delivers exponential performance, and fuses security into the network.
In December 2024, Alcatel-Lucent Enterprise introduced Fleet Supervision, a new cloud-based network inventory management solution. The new solution is designed to help organizations manage increasing network complexity, ensure software compliance, and support security needs.
In November 2024, Broadcom launched VeloRAIN (robust AI networking) architecture that uses AI and machine learning to enhance the performance and security of distributed AI workloads.
About Us:
Coherent Market Insights leads into data and analytics, audience measurement, consumer behaviours, and market trend analysis. From shorter dispatch to in-depth insights, CMI has exceled in offering research, analytics, and consumer-focused shifts for nearly a decade. With cutting-edge syndicated tools and custom-made research services, we empower businesses to move in the direction of growth. We are multifunctional in our work scope and have 450+ seasoned consultants, analysts, and researchers across 26+ industries spread out in 32+ countries.
Media Contact
Company Name: Coherent Market Insights
Contact Person: Mr Shah
Email: Send Email
Phone: 12524771362
Address:Coherent Market Insights Pvt Ltd, 533 Airport Boulevard,
City: Burlingame
State: Burlingame
Country: United States
Website: https://www.coherentmarketinsights.com/industry-reports/network-management-system-market
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Network Management System Market Forecast Predicts Significant CAGR Growth of 9.6% by 2032