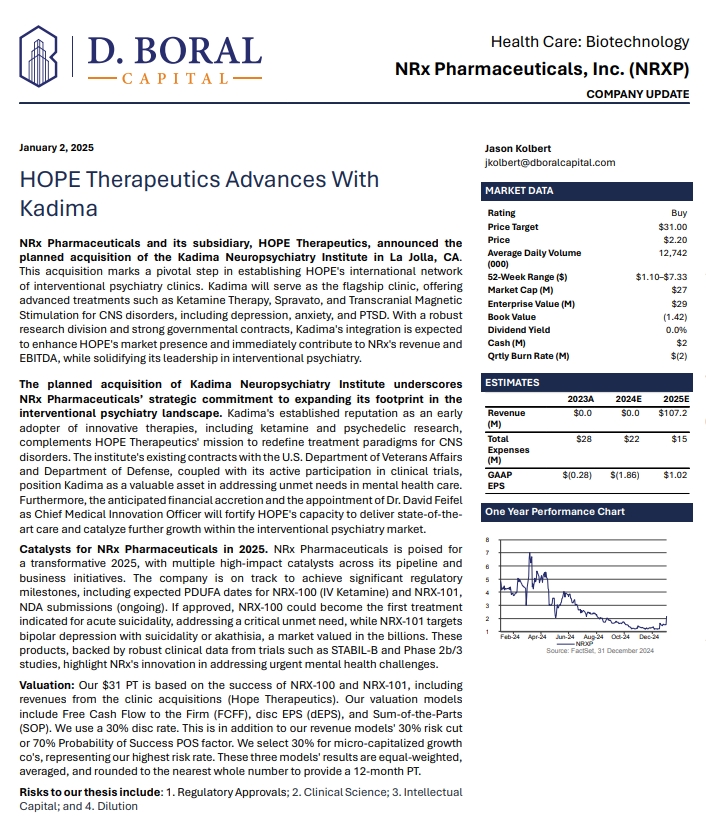
The Graves Ophthalmopathy market is expected to surge due to the disease’s increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Graves Ophthalmopathy pipeline products will significantly revolutionize the Graves Ophthalmopathy market dynamics.
DelveInsight’s “Graves Ophthalmopathy Market Insights, Epidemiology, and Market Forecast-2034″ report offers an in-depth understanding of the Graves Ophthalmopathy, historical and forecasted epidemiology as well as the Graves Ophthalmopathy market trends in the United States, EU5 (Germany, Spain, Italy, France, and United Kingdom) and Japan.
The Graves Ophthalmopathy market report covers emerging drugs, current treatment practices, market share of the individual therapies, and current & forecasted market size from 2020 to 2034. It also evaluates the current treatment practice/algorithm, market drivers & barriers, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the market.
To Know in detail about the Graves Ophthalmopathy market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Graves Ophthalmopathy Market Forecast
Some of the key facts of the Graves Ophthalmopathy Market Report:
-
The Graves Ophthalmopathy market size was valued approximately USD 2600 million in 2023 and is anticipated to grow with a significant CAGR during the study period (2020-2034)
-
In May 2025, Viridian Therapeutics, Inc. (Nasdaq: VRDN), a biopharmaceutical company dedicated to developing and commercializing potential best-in-class treatments for serious and rare conditions, has announced encouraging long-term durability results from the Phase 3 THRIVE clinical trial of veligrotug (“veli”). Veli is an intravenously administered antibody targeting the insulin-like growth factor-1 receptor (IGF-1R) and is being evaluated in patients with active thyroid eye disease (TED), an autoimmune disorder marked by inflammation, tissue expansion, and damage around and behind the eyes.
-
In January 2025, Sling Therapeutics, Inc., a late-stage biopharmaceutical company specializing in the development of oral therapies for thyroid eye disease (TED), has announced topline efficacy and safety results from its Phase 2b/3 LIDS trial evaluating linsitinib in patients with active, moderate to severe TED. Linsitinib, the company’s lead oral small molecule candidate, is administered twice daily and is currently in clinical development for TED. It targets the well-established IGF-1R pathway and has demonstrated a favorable safety profile, having been studied in over 900 patients across fifteen clinical trials in various therapeutic areas.
-
In September 2024, Viridian Therapeutics, Inc. (NASDAQ: VRDN), a biopharmaceutical company dedicated to discovering and developing potential best-in-class treatments for serious and rare diseases, today announced promising topline results from the THRIVE phase 3 clinical trial of VRDN-001, now known as veligrotug. This intravenously administered anti-insulin-like growth factor-1 receptor (IGF-1R) antibody is being evaluated in patients with active thyroid eye disease (TED), an autoimmune disorder marked by inflammation, growth, and damage to the tissues surrounding and behind the eyes
-
In 2022, approximately 2,385,900 cases of Graves Ophthalmopathy were reported in the seven major markets (7MM), and it is anticipated that these figures will rise throughout the study period from 2020 to 2034.
-
In 2022, it was estimated that there were approximately 448,800 diagnosed prevalent cases of Graves Ophthalmopathy in the United States. It is projected that these cases will increase over the study period from 2020 to 2034.
-
In 2022, Germany had the highest number of diagnosed prevalent cases of Graves Ophthalmopathy patients among the EU4 and the UK, with around 105,900 cases. Conversely, Spain had the fewest cases, with approximately 59,200 cases in 2022.
-
In the United States, the number of diagnosed prevalent cases of Graves Ophthalmopathy was higher among females compared to males in 2022, with approximately 89,800 cases in males and around 359,000 cases in females.
-
Key Graves Ophthalmopathy Companies: Immunovant Sciences GmbH, Horizon Pharma USA, Inc., Viridian Therapeutics, Inc., Tourmaline Bio, Inc., ACELYRIN Inc., Hoffmann-La Roche, Horizon Therapeutics USA, Inc., Lassen Therapeutics Inc., Novartis, Sling Therapeutics, Inc., Lassen Therapeutics, and others
-
Key Graves Ophthalmopathy Therapies: Batoclimab (IMVT 1401), RVT-1401, Teprotumumab, VRDN-001 10 mg/kg Drug, TOUR006 – 20 MG, IBI311, lonigutamab, Satralizumab, TEPEZZA, LASN01, CFZ533, linsitinib, LASN01, and others
-
The Graves Ophthalmopathy epidemiology based on gender analyzed that the females are affected more as compared to males in the case of Graves Ophthalmopathy
Graves Ophthalmopathy Overview
Graves’ ophthalmopathy, also known as thyroid eye disease or Graves’ orbitopathy, is a condition characterized by inflammation and swelling of the tissues surrounding the eyes. It is commonly associated with Graves’ disease, an autoimmune disorder that affects the thyroid gland, leading to hyperthyroidism (overactive thyroid).
Get a Free sample for the Graves Ophthalmopathy Market Report –
https://www.delveinsight.com/report-store/graves-ophthalmopathy-market
Graves Ophthalmopathy Market
The dynamics of the Graves Ophthalmopathy market are anticipated to change in the coming years owing to the expected launch of emerging therapies and others during the forecasted period 2020-2034.
“The DAVIAD project successfully resulted in the manufacture GMP grade ATX-GD-59 and completion of pharmacodynamic and toxicology studies in order to progress the therapeutic vaccine into a Phase I/IIa clinical trial. In the clinical trial, the vaccine was proven to be safe and well tolerated.”
Graves Ophthalmopathy Epidemiology
The epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Graves Ophthalmopathy Epidemiology Segmentation:
The Graves Ophthalmopathy market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
-
Prevalent Cases of Graves Ophthalmopathy in the 7MM
-
Diagnosed Prevalent Cases of Graves Ophthalmopathy in the 7MM
-
Gender-specific Diagnosed Prevalent Cases of Graves Ophthalmopathy in the 7MM
-
Moderate-to-severe Drug-treated Cases of Acute Graves Ophthalmopathy in the 7MM
-
Acute Diagnosed Prevalent Cases of Graves Ophthalmopathy in the 7MM
-
Diagnosed Prevalent Cases of Graves Ophthalmopathy by Chronicity in the 7MM
Download the report to understand which factors are driving Graves Ophthalmopathy epidemiology trends @ Graves Ophthalmopathy Epidemiological Insights
Graves Ophthalmopathy Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Graves Ophthalmopathy market or expected to get launched during the study period. The analysis covers Graves Ophthalmopathy market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Graves Ophthalmopathy Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Graves Ophthalmopathy Therapies and Key Companies
-
Batoclimab (IMVT 1401): Immunovant Sciences
-
RVT-1401: Immunovant Sciences GmbH
-
Teprotumumab: Horizon Pharma USA, Inc.
-
VRDN-001 10 mg/kg Drug: Viridian Therapeutics, Inc.
-
Batoclimab: Immunovant Sciences GmbH
-
TOUR006 – 20 MG: Tourmaline Bio, Inc.
-
IBI311: Innovent Biologics (Suzhou) Co. Ltd.
-
lonigutamab: ACELYRIN Inc.
-
Satralizumab: Hoffmann-La Roche
-
TEPEZZA: Horizon Therapeutics USA, Inc.
-
LASN01: Lassen Therapeutics Inc.
-
CFZ533: Novartis
-
linsitinib: Sling Therapeutics, Inc.
-
LASN01: Lassen Therapeutics
To know more about Graves Ophthalmopathy treatment, visit @ Graves Ophthalmopathy Medications
Graves Ophthalmopathy Market Strengths
-
The recent US FDA approval of Horizon’s TEPEZZA has accelerated the research and development of GO.
-
Recent efforts to increase patient and clinician awareness of GO, with various awareness campaigns, are expected to increase early diagnosis and treatment of GO.
Graves Ophthalmopathy Market opportunities
-
Designing an effective molecule with an intended delivery mode that is matched to the intended target compartment is a necessary first step in ocular drug discovery programs.
-
Lack of availability of disease-modifying treatment for chronic patients, who have a significant impact on their emotional and social well-being.
Scope of the Graves Ophthalmopathy Market Report
-
Study Period: 2020–2034
-
Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
-
Key Graves Ophthalmopathy Companies: Novartis, AdvanceCor, Apitope, Worg Pharmaceuticals, AV7 Limited, and others
-
Key Graves Ophthalmopathy Therapies: Batoclimab (IMVT 1401), RVT-1401, Teprotumumab, VRDN-001 10 mg/kg Drug, TOUR006 – 20 MG, IBI311, lonigutamab, Satralizumab, TEPEZZA, LASN01, CFZ533, linsitinib, LASN01, and others
-
Graves Ophthalmopathy Therapeutic Assessment: Graves Ophthalmopathy current marketed and Graves Ophthalmopathy emerging therapies
-
Graves Ophthalmopathy Market Dynamics: Graves Ophthalmopathy market drivers and Graves Ophthalmopathy market barriers
-
Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
-
Graves Ophthalmopathy Unmet Needs, KOL’s views, Analyst’s views, Graves Ophthalmopathy Market Access and Reimbursement
Discover more about therapies set to grab major Graves Ophthalmopathy market share @ Graves Ophthalmopathy Treatment Landscape
Table of Contents
1. Graves Ophthalmopathy Market Report Introduction
2. Executive Summary for Graves Ophthalmopathy
3. SWOT analysis of Graves Ophthalmopathy
4. Graves Ophthalmopathy Patient Share (%) Overview at a Glance
5. Graves Ophthalmopathy Market Overview at a Glance
6. Graves Ophthalmopathy Disease Background and Overview
7. Graves Ophthalmopathy Epidemiology and Patient Population
8. Country-Specific Patient Population of Graves Ophthalmopathy
9. Graves Ophthalmopathy Current Treatment and Medical Practices
10. Graves Ophthalmopathy Unmet Needs
11. Graves Ophthalmopathy Emerging Therapies
12. Graves Ophthalmopathy Market Outlook
13. Country-Wise Graves Ophthalmopathy Market Analysis (2020–2034)
14. Graves Ophthalmopathy Market Access and Reimbursement of Therapies
15. Graves Ophthalmopathy Market Drivers
16. Graves Ophthalmopathy Market Barriers
17. Graves Ophthalmopathy Appendix
18. Graves Ophthalmopathy Report Methodology
19. DelveInsight Capabilities
20. Disclaimer
21. About DelveInsight
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Media Contact
Company Name: DelveInsight
Contact Person: Gaurav Bora
Email: Send Email
Phone: +14699457679
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Graves Ophthalmopathy Market Set to Grow Substantially Through 2034, DelveInsight Projects | Novartis, AdvanceCor, Apitope, Worg Pharma, AV7 Limited