
In a world where technology is everywhere and screen time is often met with guilt, one app is flipping the narrative for families. Meet Kreebo, the AI-powered storytelling companion built especially for children—a magical space where imagination blossoms, creativity flows freely, and screen time becomes something to celebrate.
Kreebo officially launches today, promising something rare and powerful: a safe, child friendly AI experience that helps kids bring their wildest ideas to life—one story at a time.
Because every child has a story to tell.
A Parent’s Problem Turned Into a Global Solution
Kreebo’s story began with one father’s heartbreak.
“My 8-year-old son had incredible ideas—whole worlds he’d imagine during bedtime or car rides,” said the founder of Kreebo. “But when it came time to draw or write them, he’d struggle. His thoughts were vivid, but his words couldn’t keep up. He got frustrated. I searched everywhere for a tool that could help him turn those ideas into something real—and I couldn’t find one.”
So, he built it.
Kreebo was born to bridge that gap: a magical AI friend who listens to kids, chats with them, and helps turn their ideas into beautifully illustrated storybooks. No pressure. No perfection required. Just imagination—and a safe space to grow it.

What Makes Kreebo Different?
Kreebo isn’t like the tech tools parents are used to. There are no games filled with ads, no endless scrolling, and no algorithms mining data.
What children get instead is:
• A guided storytelling experience through friendly AI chat
• A way to co-create storybooks they can be proud of
• A tool that’s age-appropriate, ad-free, and safe
• Illustrated books they can read, share, and revisit
• A feeling of confidence and joy that comes from being heard
Unlike generic apps, Kreebo was designed hand-in-hand with parents, teachers, illustrators, and child development experts. It respects children’s emotional world—and protects it.
“Kreebo is the one time I don’t feel guilty about screen time,” said one mother of two. “My daughter’s not just watching cartoons. She’s creating worlds. And she’s so proud of what she makes.”

The Science Behind the Magic
According to Dr. John Medina, a developmental molecular biologist and author of Brain Rules for Baby, early childhood is marked by an extraordinary burst in neural connections.
“Children’s brains are wired for learning and creativity,” he explains. “During early development, they form more neural connections than at any other time in life. This is what gives young kids that boundless potential.”
However, without stimulation, those connections are pruned away. The brain becomes more specialized, but it also loses some of its raw creative power.
That’s why Kreebo matters.
By giving kids the space, encouragement, and tools to create freely, Kreebo helps them explore their ‘genius’ phase—turning fleeting ideas into something lasting and meaningful.
“We can’t stop time,” says the founder. “But we can make sure that while they’re young, children have a platform to express their true, unfiltered imagination. And that’s what Kreebo gives them.”
Kids Don’t Just Play with Kreebo. They Create.
Children as young as six are now publishing books through Kreebo.
They write about alien cupcakes, lost puppies in New York City, magical forests guarded by talking owls, and superheroes who wear glittery sneakers. Kreebo listens patiently, offers prompts when they’re stuck, and celebrates every twist and turn of their story.
And then? It brings those tales to life—with vibrant illustrations and a beautifully formatted digital book they can read, print, or share.
Some of Kreebo’s young authors have already created multiple books. For many, it’s their first time seeing themselves as storytellers.
“I made a book! Like a real book!” exclaimed an excited 7-year-old user.
“I showed it to my grandma and she cried.”

Built with Care. Backed by Purpose.
Kreebo’s team is made up of designers, educators, and parents who understand the delicate balance of screen time and meaningful engagement. They know how hard it is to find tech that doesn’t just occupy a child’s time—but enriches it.
“Our mission is simple: to give kids something good on their screens,” says co-founder Saraf Talukder. “Something that feeds their imagination instead of draining it.”
And because safety is non-negotiable:
• Kreebo never exposes kids to open web content
• There are no in-app purchases, pop-ups, or hidden costs
• Conversations are guided, moderated, and age-aligned
• Stories are created in a closed, protected environment
A Platorm for Every Child
Whether a child is 6, 8, or 10, Kreebo meets them where they are. Whether they love writing or dread it. Whether they’re brimming with ideas or just need a nudge. Every child gets to be an author.
For parents, it’s a way to turn downtime into something they can be proud of.
“I’ve tried so many apps for my kids,” shared one father. “Most of them are either too complicated or too mindless. But Kreebo hits that perfect balance. My son is talking to an AI— but it feels like talking to a friend who helps him dream bigger.”
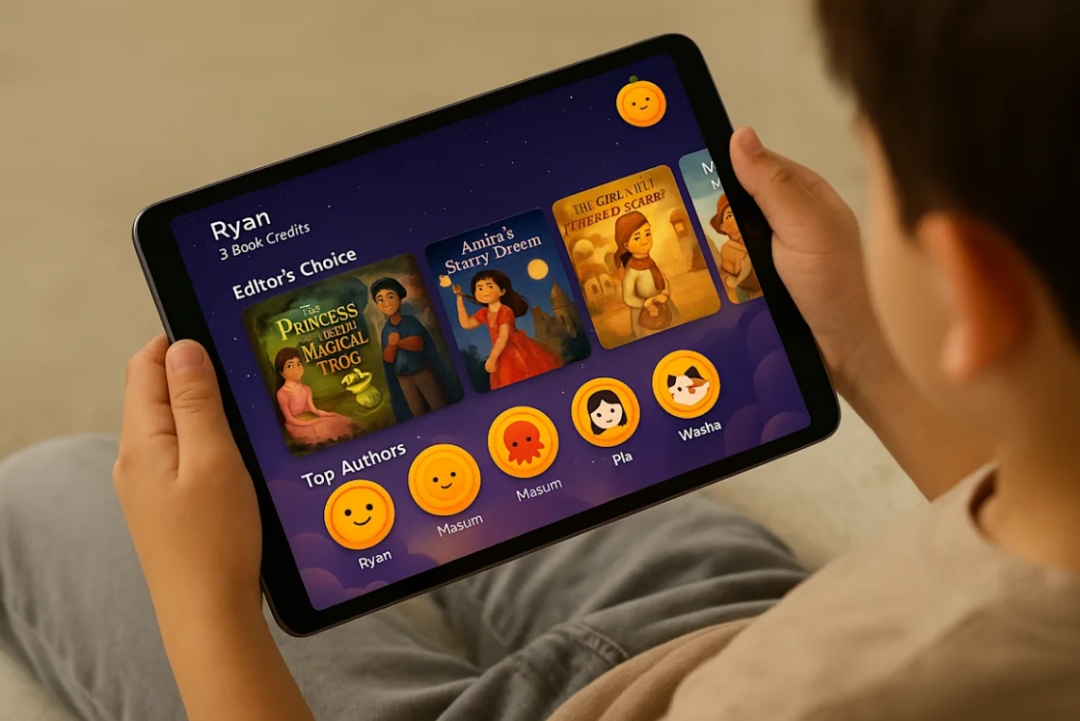
The Future of Storytelling Starts Here
Kreebo is more than just an app. It’s a movement toward joyful, safe, purposeful tech for children.
It proves that AI, when used mindfully, can nurture the most human thing of all: imagination.
What started as a solution for one child is now a platform built for every child. Because every child has a story to tell, Kreebo just helps them tell it.
ABOUT KREEBO
Kreebo is a child-friendly AI storytelling platform co-founded by Saraf Talukder and Wahid Choudhury, designed to help children turn their imagination into beautifully illustrated books. Built with safety, creativity, and education in mind, Kreebo transforms screen time into story time—without ads, distractions, or pressure. Through a playful, guided chat experience, kids become proud authors while parents finally have a screen activity they can feel good about.
Learn more at: www.kreebo.com
Download the App:
iOS: https://apps.apple.com/us/app/kreebo/id6743721247
Android: https://play.google.com/store/apps/details?id=com.gurho.kreebo
Media Contact
Company Name: Kreebo
Contact Person: Saraf Talukder
Email: Send Email
Country: United States
Website: www.kreebo.com













