DelveInsight’s, “Complicated Urinary Tract Infections Pipeline Insight 2025” report provides comprehensive insights about 12+ companies and 12+ pipeline drugs in Complicated Urinary Tract Infections pipeline landscape. It covers the Complicated Urinary Tract Infection pipeline drug profiles, including clinical and nonclinical stage products. It also covers the therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Stay ahead with the latest insights! Download DelveInsight’s comprehensive Complicated Urinary Tract Infection Pipeline Report to explore emerging therapies, key players, and future treatment landscapes @ Complicated Urinary Tract Infection Pipeline Outlook Report
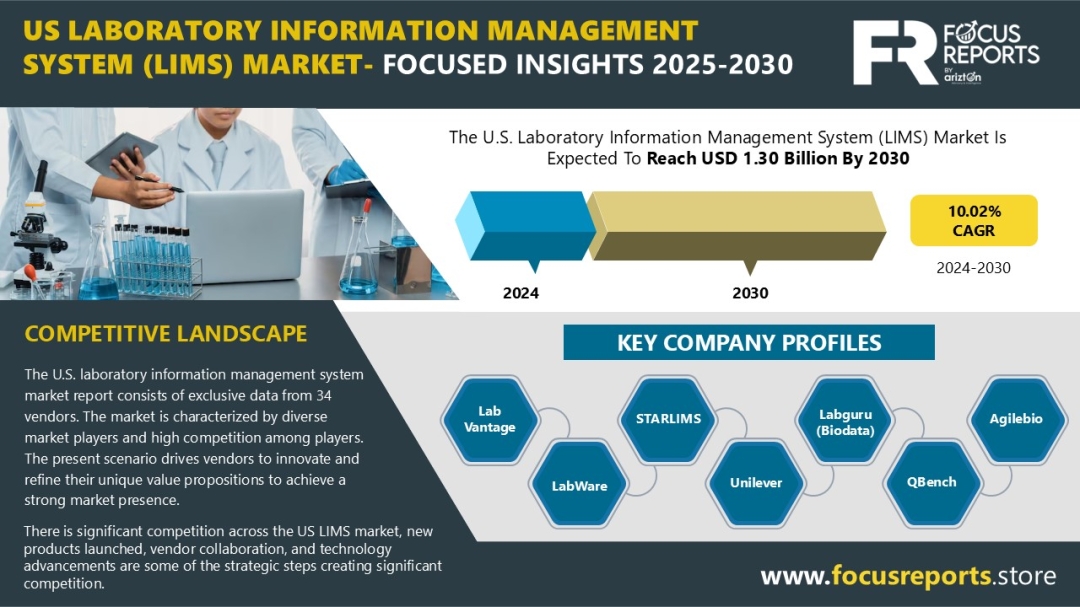
Key Takeaways from the Complicated Urinary Tract Infection Pipeline Report
- In June 2025, Menarini Group announced a clinical trial is to assess the pharmacokinetic (PK) and safety and tolerability of Vaborem ( fixed combination of meropenem and vaborbactam) in the paediatric population aged from 3 months to < 18 years with complicated urinary tract infection (cUTI) including acute pyelonephritis (AP) in need of hospitalisation and intravenous (IV) antibiotic administration.
- In June 2025, Venatorx Pharmaceuticals Inc. conducted a Phase 3 study Evaluating the Efficacy, Safety, and Tolerability of Cefepime/VNRX-5133 in Adults With Complicated Urinary Tract Infections (cUTI), Including Acute Pyelonephritis.
- In June 2025, Rempex organized a study is to assess the safety and tolerability of meropenem-vaborbactam administered by intravenous (IV) infusion in children 3 months and above to less than 12 years with complicated urinary tract infections (cUTI), including acute pyelonephritis (AP).
- DelveInsight’s Complicated Urinary Tract Infection pipeline report depicts a robust space with 12+ active players working to develop 12+ pipeline therapies for Complicated Urinary Tract Infection treatment.
- The leading Complicated Urinary Tract Infection Companies such as Spero Therapeutics, Iterum Therapeutics, Venatorx Pharmaceuticals Inc., Nabriva Therapeutics, Allecra Therapeutics, Wockhardt, MerLion Pharmaceuticals, and others.
- Promising Complicated Urinary Tract Infections Pipeline Therapies such as Ceftazidime -avibactam, Cefepime, CXA-101, Ceftazidime, Eravacycline, Ertapenem, Sulopenem and others.
Discover how the Complicated Urinary Tract Infection treatment paradigm is evolving. Access DelveInsight’s in-depth Complicated Urinary Tract Infection Pipeline Analysis for a closer look at promising breakthroughs @ Complicated Urinary Tract Infection Clinical Trials and Studies
Complicated Urinary Tract Infection Emerging Drugs Profile
- Tebipenem Pivoxil Hydrobromide: Spero Therapeutics
Spero Therapeutics is developing tebipenem HBr (tebipenem pivoxil hydrobromide; formerly SPR994) as an oral antibiotic for the treatment of complicated urinary tract infection (cUTI) and acute pyelonephritis (AP) to help patients avoid hospitalizations (stay at home) and/or transition patients home after IV therapy (get home). In September 2020, tebipenem HBr completed a pivotal Phase III trial, ADAPT-PO, for the treatment of cUTI, including acute pyelonephritis (AP). ADAPT-PO is a landmark trial that is the first ever to test an all oral regimen against an all intravenous (IV) regimen for the treatment of cUTI. The global, randomized, placebo-controlled ADAPT-PO trial evaluated the safety and efficacy of tebipenem HBr in hospitalized adult patients with cUTI or AP. Data from the trial demonstrated that oral tebipenem HBr was statistically non-inferior to IV ertapenem in the treatment of patients with cUTI and patients with AP. If approved, tebipenem HBr would be the first oral carbapenem antimicrobial to receive marketing approval in the United States. Tebipenem HBr has been granted Qualified Infectious Disease Product (QIDP) and Fast Track designations by the FDA for the treatment of cUTI and AP.Following feedback from the US FDA at Spero’s recent Type A meeting, Spero will conduct an additional phase III trial to support the regulatory submission.
- Sulopenem: Iterum Therapeutics
Sulopenem is an orally bioavailable, broad-spectrum penem β-lactam antibiotic which is being developed for the treatment of infections caused by multi-drug resistant bacteria. Sulopenem was discovered in the laboratories of Pfizer Inc. in the 1980s and was first developed with an intravenous (IV) formulation only. The company completed an extensive pre-clinical program, followed by human studies. Later, an oral formulation was developed and tested in Phase I and Phase II trials. In May 2022, Iterum Therapeutics plc announced it met with the US Food and Drug Administration (“FDA”) on May 5, 2022, to discuss its proposed plan to conduct one additional Phase 3 clinical trial to support the potential resubmission of the Company’s new drug application (“NDA”) for oral sulopenem etzadroxil-probenecid (“oral sulopenem”) for the treatment of uncomplicated urinary tract infections (“uUTI”). Currently, the drug is in Phase III stage of clinical trial evaluation for the treatment uncomplicated urinary tract infections.
The Complicated Urinary Tract Infection Pipeline Report Provides Insights into
- The report provides detailed insights about companies that are developing therapies for the treatment of Complicated Urinary Tract Infection with aggregate therapies developed by each company for the same.
- It accesses the Different therapeutic candidates segmented into early-stage, mid-stage, and late-stage of development for Complicated Urinary Tract Infection Treatment.
- Complicated Urinary Tract Infection Companies are involved in targeted therapeutics development with respective active and inactive (dormant or discontinued) projects.
- Complicated Urinary Tract Infection Drugs under development based on the stage of development, route of administration, target receptor, monotherapy or combination therapy, a different mechanism of action, and molecular type.
- Detailed analysis of collaborations (company-company collaborations and company-academia collaborations), licensing agreement and financing details for future advancement of the Complicated Urinary Tract Infection market
Explore groundbreaking therapies and clinical trials in the Complicated Urinary Tract Infection Pipeline. Access DelveInsight’s detailed report now! @ New Complicated Urinary Tract Infection Drugs
Complicated Urinary Tract Infection Companies
Spero Therapeutics, Iterum Therapeutics, Venatorx Pharmaceuticals Inc., Nabriva Therapeutics, Allecra Therapeutics, Wockhardt, MerLion Pharmaceuticals, and others
Complicated Urinary Tract Infections pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration. Products have been categorized under various ROAs such as
- Oral
- Parenteral
- Intravenous
- Subcutaneous
- Topical
Complicated Urinary Tract Infection Products have been categorized under various Molecule types such as
- Monoclonal Antibody
- Peptides
- Polymer
- Small molecule
- Gene therapy
Download DelveInsight’s latest report to gain strategic insights into upcoming therapies and key developments @ Complicated Urinary Tract Infection Market Drivers and Barriers, and Future Perspectives
Scope of the Complicated Urinary Tract Infection Pipeline Report
- Coverage- Global
- Complicated Urinary Tract Infection Companies- Spero Therapeutics, Iterum Therapeutics, Venatorx Pharmaceuticals Inc., Nabriva Therapeutics, Allecra Therapeutics, Wockhardt, MerLion Pharmaceuticals, and others.
- Complicated Urinary Tract Infections Pipeline Therapies- Ceftazidime -avibactam, Cefepime, CXA-101, Ceftazidime, Eravacycline, Ertapenem, Sulopenem and others.
- Complicated Urinary Tract Infection Therapeutic Assessment by Product Type: Mono, Combination, Mono/Combination
- Complicated Urinary Tract Infection Therapeutic Assessment by Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
Which companies are leading the race in Complicated Urinary Tract Infection drug development? Find out in DelveInsight’s exclusive Complicated Urinary Tract Infection Pipeline Report—access it now! @ Complicated Urinary Tract Infection Emerging Drugs and Major Companies
Table of Contents
- Introduction
- Executive Summary
- Complicated Urinary Tract Infections: Overview
- Pipeline Therapeutics
- Therapeutic Assessment
- Late Stage Products (Phase III)
- Tebipenem Pivoxil Hydrobromide: Spero Therapeutics
- Drug profiles in the detailed report…..
- Mid Stage Products (Phase II)
- Drug Name: Company Name
- Drug profiles in the detailed report…..
- Early stage products (Phase I)
- ETX0282CPDP: Entasis Therapeutics
- Drug profiles in the detailed report…..
- Inactive Products
- Complicated Urinary Tract Infections Key Companies
- Complicated Urinary Tract Infections Key Products
- Complicated Urinary Tract Infections- Unmet Needs
- Complicated Urinary Tract Infections- Market Drivers and Barriers
- Complicated Urinary Tract Infections- Future Perspectives and Conclusion
- Complicated Urinary Tract Infections Analyst Views
- Complicated Urinary Tract Infections Key Companies
- Appendix
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Yash Bhardwaj
Email: Send Email
Phone: 09650213330
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/report-store/complicated-urinary-tract-infections-pipeline-insight
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Complicated Urinary Tract Infection Pipeline Appears Robust With 12+ Key Pharma Companies Actively Working in the Therapeutics Segment | DelveInsight