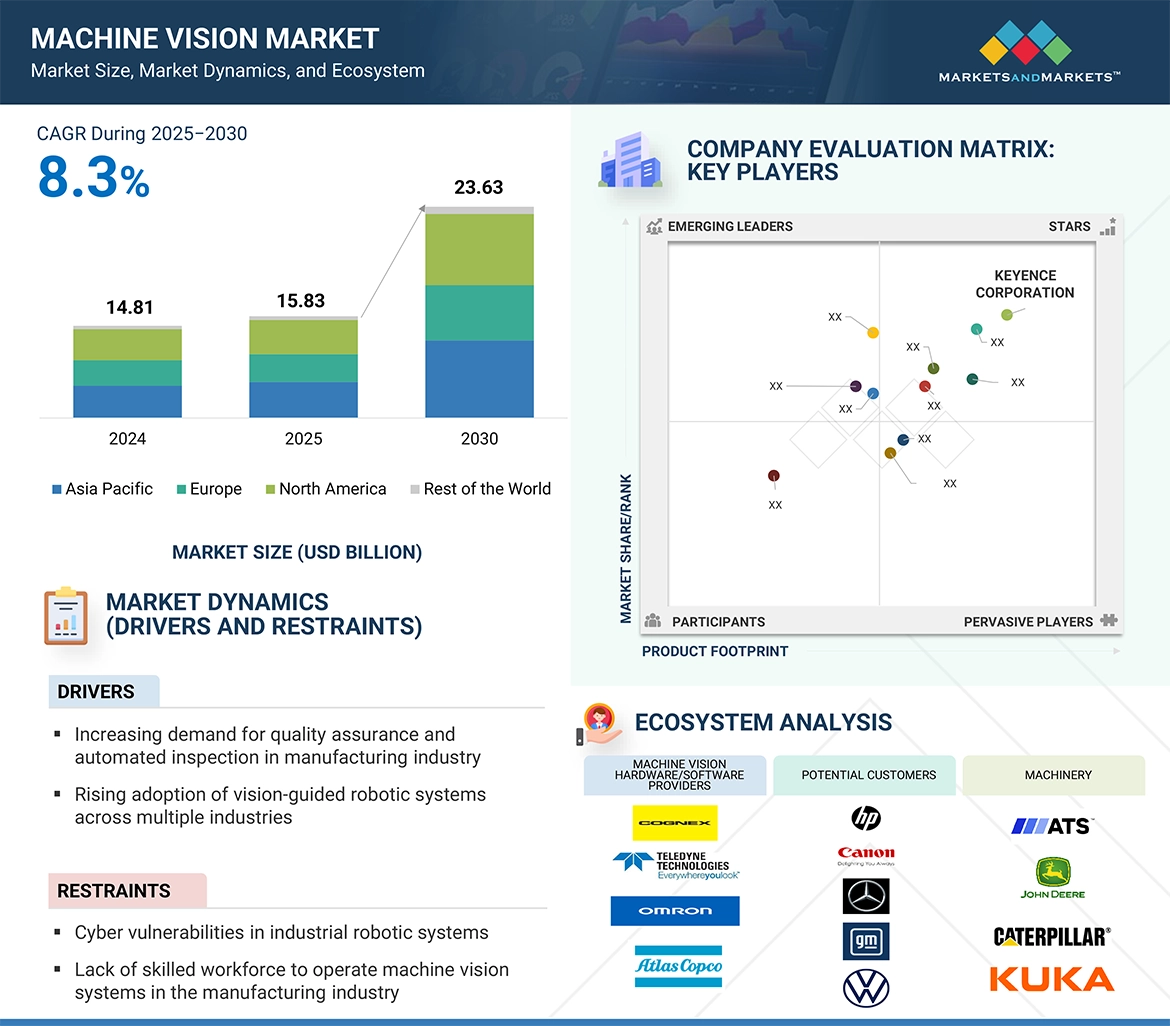
DelveInsight’s, “Diffuse Large B-Cell Lymphoma Pipeline Insight 2025” report provides comprehensive insights about 70+ companies and 75+ pipeline drugs in Diffuse Large B-Cell Lymphoma pipeline landscape. It covers the Diffuse Large B-Cell Lymphoma pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Diffuse Large B-Cell Lymphoma therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive Diffuse Large B-Cell Lymphoma pipeline products in this space.
Stay ahead with the latest insights! Download DelveInsight’s comprehensive Diffuse Large B-Cell Lymphoma Pipeline Report to explore emerging therapies, key Diffuse Large B-Cell Lymphoma Companies, and future Diffuse Large B-Cell Lymphoma treatment landscapes @ Diffuse Large B-Cell Lymphoma Pipeline Outlook Report
Key Takeaways from the Diffuse Large B-Cell Lymphoma Pipeline Report
- In June 2025, Genmab announced a study will assess how safe and effective epcoritamab plus lenalidomide (E-Len) is compared to rituximab plus gemcitabine and oxaliplatin (R-GemOx) )in treating adult participants with relapsed or refractory (R/R) Diffuse Large B-Cell Lymphoma (DLBCL). Adverse events and change in disease condition will be assessed.
- In June 2025, Mitsubishi Tanabe Pharma Corporation conducted a phase I/II Open-Label Study of MT-2111 in Patients With Relapsed/Refractory DLBCL.
- DelveInsight’s Diffuse Large B-Cell Lymphoma pipeline report depicts a robust space with 70+ active players working to develop 75+ pipeline therapies for Diffuse Large B-Cell Lymphoma treatment.
- The leading Diffuse Large B-Cell Lymphoma Companies such as Miltenyi Biomedicine, Adicet Bio, VelosBio, Novartis Pharmaceuticals, Sanofi, Eisai Co, Schrodinger, Sana Biotechnology, Ranok Therapeutics, Monte Rosa Therapeutic, Otsuka Pharmaceutical, OncoNano Medicine, Regeneron Pharmaceuticals, Hoffmann-La Roche, Celgene, Nurix Therapeutics, NovalGen, Nektar Therapeutics, Genentech, CSPC ZhongQi Pharmaceutical Technology and others.
- Promising Diffuse Large B-Cell Lymphoma Therapies such as Ibrutinib, Bendamustine, Rituximab, R-miniCHOP, Selinexor, Tafasitamab, Lenalidomide, and others.
Discover how the Diffuse Large B-Cell Lymphoma treatment paradigm is evolving. Access DelveInsight’s in-depth Diffuse Large B-Cell Lymphoma Pipeline Analysis for a closer look at promising breakthroughs @ Diffuse Large B-Cell Lymphoma Clinical Trials and Studies
Diffuse Large B-Cell Lymphoma Emerging Drugs Profile
- Brentuximab vedotin: Pfizer
Brentuximab vedotin (Adcetris) is an anti-neoplastic agent. It is indicated for the treatment of patients with Classical Hodgkin lymphoma (HL) after failure of autologous hematopoietic stem cell transplantation (auto-HSCT) or after failure of at least two prior multi-agent chemotherapy regimens in patients who are not auto-HSCT candidates. Adcetris is also indicated for the treatment of adult patients with CD30+ Hodgkin lymphoma at increased risk of relapse or progression following autologous stem cell transplant (ASCT), also indicated for the treatment of adult patients with relapsed or refractory systemic anaplastic large cell lymphoma, for the treatment of adult patients with primary cutaneous anaplastic large cell lymphoma (pcALCL) and CD30-expressing mycosis fungoides (MF) who have received prior systemic therapy, for the treatment of adult patients with relapsed or refractory CD30+ cutaneous T- cell lymphoma (CTCL) after at least 1 prior systemic therapy. Adcetris is indicated for the first-line pediatric treatment for CD30-positive Hodgkin lymphoma. Currently, the drug is in Phase III stage of its clinical trial for the treatment of Diffuse Large B Cell Lymphoma.
- THOR-707: Sanofi
THOR-707 is a precisely PEGylated version of IL-2, where the PEG chain is attached to a novel amino acid inserted at a location on IL-2 that prevents it from engaging the alpha-receptor and binding to immune receptors that cause drug toxicities (IL-2R-alpha, CD25). The engineered IL-2 retains near-native binding to the beta-gamma receptors that selectively expand tumor-killing T effector cells and Natural Killer (NK) cells without the alpha-mediated immunosuppressive effects of regulatory T cells or eosinophil-mediated vascular leak syndrome. Currently, the drug is in Phase II stage of its clinical trial for the treatment of Diffuse Large B Cell Lymphoma.
- Abexinostat: Xynomic Pharmaceuticals
Abexinostat (Xynomic Pharmaceuticals) is a broad histone deacetylase (HDAC) inhibitor. HDAC enzymes (also known as lysine deacetylase) cleave acetyl groups from N-acetyl lysine amino acids on a histone. HDAC inhibition leads to highly acetylated histones and chromatin reshaping. In addition to altering histone acetylation, HDAC inhibitors can also influence the degree of acetylation on non-histone proteins, increasing or repressing their activity. Currently, the drug is in Phase II stage of its clinical trial for the treatment of Diffuse Large B-cell lymphoma.
- RNK05047: Ranok Therapeutics
RNK05047 is a first-in-class, small-molecule, tumor- and BRD4-selective protein degrader that was discovered and developed using Ranok’s proprietary approach to targeted protein degradation, CHAMPTM. The bromodomain transcription factor BRD4 is a key regulator of oncogenes such as MYC and BCL2 and is involved in diverse cancer types. CHAMP-1 is a Phase I/II trial of RNK05047 currently underway in the US that will assess its safety, tolerability, and pharmacokinetics, and also includes measures of anti-tumor activity and pharmacodynamics readouts as secondary endpoints.
- BMF-219: Biomea Fusion
BMF-219 is an oral investigational covalent menin inhibitor. Data suggests that optimized covalent inhibitors can provide deeper inhibition while being better tolerated than some conventional reversible inhibitors. BMF-219 is being developed for genetically defined AML, ALL, DLBCL, MM and CLL patients. BMF-219 blocks the interaction of menin and MLL (AML, ALL), and limits the activity and/or expression of NPM1, MYC, HOX, and MEIS1, all known drivers of oncogenic proliferation and survival. Currently, the drug is in Phase I stage of its clinical trial for the treatment of Diffuse Large B Cell Lymphoma.
- ADI-001: Adicet Bio
ADI-001 is an investigational allogeneic gamma delta CAR T cell therapy being developed as a potential treatment for relapsed or refractory B-cell NHL. ADI-001 targets malignant B-cells via an anti-CD20 CAR and via the gamma delta innate and T cell endogenous cytotoxicity receptors. Gamma delta T cells engineered with an anti-CD20 CAR have demonstrated potent anti-tumor activity in preclinical models, leading to long-term control of tumor growth. In April 2022, ADI-001 was granted Fast Track Designation by the FDA for the potential treatment of relapsed or refractory B-cell NHL. Currently, the drug is in Phase I stage of its clinical trial for the treatment of Diffuse Large B Cell Lymphoma.
The Diffuse Large B-Cell Lymphoma pipeline report provides insights into
- The report provides detailed insights about companies that are developing therapies for the treatment of Diffuse Large B-Cell Lymphoma with aggregate therapies developed by each company for the same.
- It accesses the Different therapeutic candidates segmented into early-stage, mid-stage, and late-stage of development for Diffuse Large B-Cell Lymphoma Treatment.
- Diffuse Large B-Cell Lymphoma Companies are involved in targeted therapeutics development with respective active and inactive (dormant or discontinued) projects.
- Diffuse Large B-Cell Lymphoma Drugs under development based on the stage of development, route of administration, target receptor, monotherapy or combination therapy, a different mechanism of action, and molecular type.
- Detailed analysis of collaborations (company-company collaborations and company-academia collaborations), licensing agreement and financing details for future advancement of the Diffuse Large B-Cell Lymphoma market.
Get a detailed analysis of the latest innovations in the Diffuse Large B-Cell Lymphoma pipeline. Explore DelveInsight’s expert-driven report today! @ Diffuse Large B-Cell Lymphoma Unmet Needs
Diffuse Large B-Cell Lymphoma Companies
Miltenyi Biomedicine, Adicet Bio, VelosBio, Novartis Pharmaceuticals, Sanofi, Eisai Co, Schrodinger, Sana Biotechnology, Ranok Therapeutics, Monte Rosa Therapeutic, Otsuka Pharmaceutical, OncoNano Medicine, Regeneron Pharmaceuticals, Hoffmann-La Roche, Celgene, Nurix Therapeutics, NovalGen, Nektar Therapeutics, Genentech, CSPC ZhongQi Pharmaceutical Technology and others.
Diffuse Large B-Cell Lymphoma pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration. Products have been categorized under various ROAs such as
- Oral
- Intravenous
- Subcutaneous
- Parenteral
- Topical
Diffuse Large B-Cell Lymphoma Products have been categorized under various Molecule types such as
- Recombinant fusion proteins
- Small molecule
- Monoclonal antibody
- Peptide
- Polymer
- Gene therapy
Download DelveInsight’s latest report to gain strategic insights into upcoming Diffuse Large B-Cell Lymphoma Therapies and key Diffuse Large B-Cell Lymphoma Developments @ Diffuse Large B-Cell Lymphoma Market Drivers and Barriers, and Future Perspectives
Scope of the Diffuse Large B-Cell Lymphoma Pipeline Report
- Coverage- Global
- Diffuse Large B-Cell Lymphoma Companies- Miltenyi Biomedicine, Adicet Bio, VelosBio, Novartis Pharmaceuticals, Sanofi, Eisai Co, Schrodinger, Sana Biotechnology, Ranok Therapeutics, Monte Rosa Therapeutic, Otsuka Pharmaceutical, OncoNano Medicine, Regeneron Pharmaceuticals, Hoffmann-La Roche, Celgene, Nurix Therapeutics, NovalGen, Nektar Therapeutics, Genentech, CSPC ZhongQi Pharmaceutical Technology and others.
- Diffuse Large B-Cell Lymphoma Therapies– Ibrutinib, Bendamustine, Rituximab, R-miniCHOP, Selinexor, Tafasitamab, Lenalidomide, and others.
- Diffuse Large B-Cell Lymphoma Therapeutic Assessment by Product Type: Mono, Combination, Mono/Combination
- Diffuse Large B-Cell Lymphoma Therapeutic Assessment by Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
Which companies are leading the race in Diffuse Large B-Cell Lymphoma drug development? Find out in DelveInsight’s exclusive Diffuse Large B-Cell Lymphoma Pipeline Report—access it now! @ Diffuse Large B-Cell Lymphoma Emerging Drugs and Major Companies
Table of Content
- Introduction
- Executive Summary
- Diffuse Large B-Cell Lymphoma: Overview
- Pipeline Therapeutics
- Therapeutic Assessment
- Diffuse Large B-Cell Lymphoma– DelveInsight’s Analytical Perspective
- Late Stage Products (Phase III)
- Brentuximab vedotin: Pfizer
- Drug profiles in the detailed report…..
- Mid Stage Products (Phase II)
- THOR-707: Sanofi
- Drug profiles in the detailed report…..
- Early Stage Products (Phase I)
- ADI-001: Adicet Bio
- Drug profiles in the detailed report…..
- Preclinical and Discovery Stage Products
- Drug name: Company name
- Drug profiles in the detailed report…..
- Inactive Products
- Diffuse Large B-Cell Lymphoma Key Companies
- Diffuse Large B-Cell Lymphoma Key Products
- Diffuse Large B-Cell Lymphoma- Unmet Needs
- Diffuse Large B-Cell Lymphoma- Market Drivers and Barriers
- Diffuse Large B-Cell Lymphoma- Future Perspectives and Conclusion
- Diffuse Large B-Cell Lymphoma Analyst Views
- Diffuse Large B-Cell Lymphoma Key Companies
- Appendix
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Yash Bhardwaj
Email: Send Email
Phone: 09650213330
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/report-store/diffuse-large-b-cell-lymphoma-pipeline-insight
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Diffuse Large B-Cell Clinical Trial Analysis: Key Insights into Rich Pipeline Featuring 70+ Companies and 75+ Therapies | DelveInsight