DelveInsight’s, “Basal Cell Carcinoma Pipeline Insight 2025” report provides comprehensive insights about 25+ companies and 25+ pipeline drugs in Basal Cell Carcinoma pipeline landscape. It covers the Basal Cell Carcinoma pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Basal Cell Carcinoma pipeline therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Explore our latest breakthroughs in Basal Cell Carcinoma Research. Learn more about our innovative pipeline today! @ Basal Cell Carcinoma Pipeline Outlook
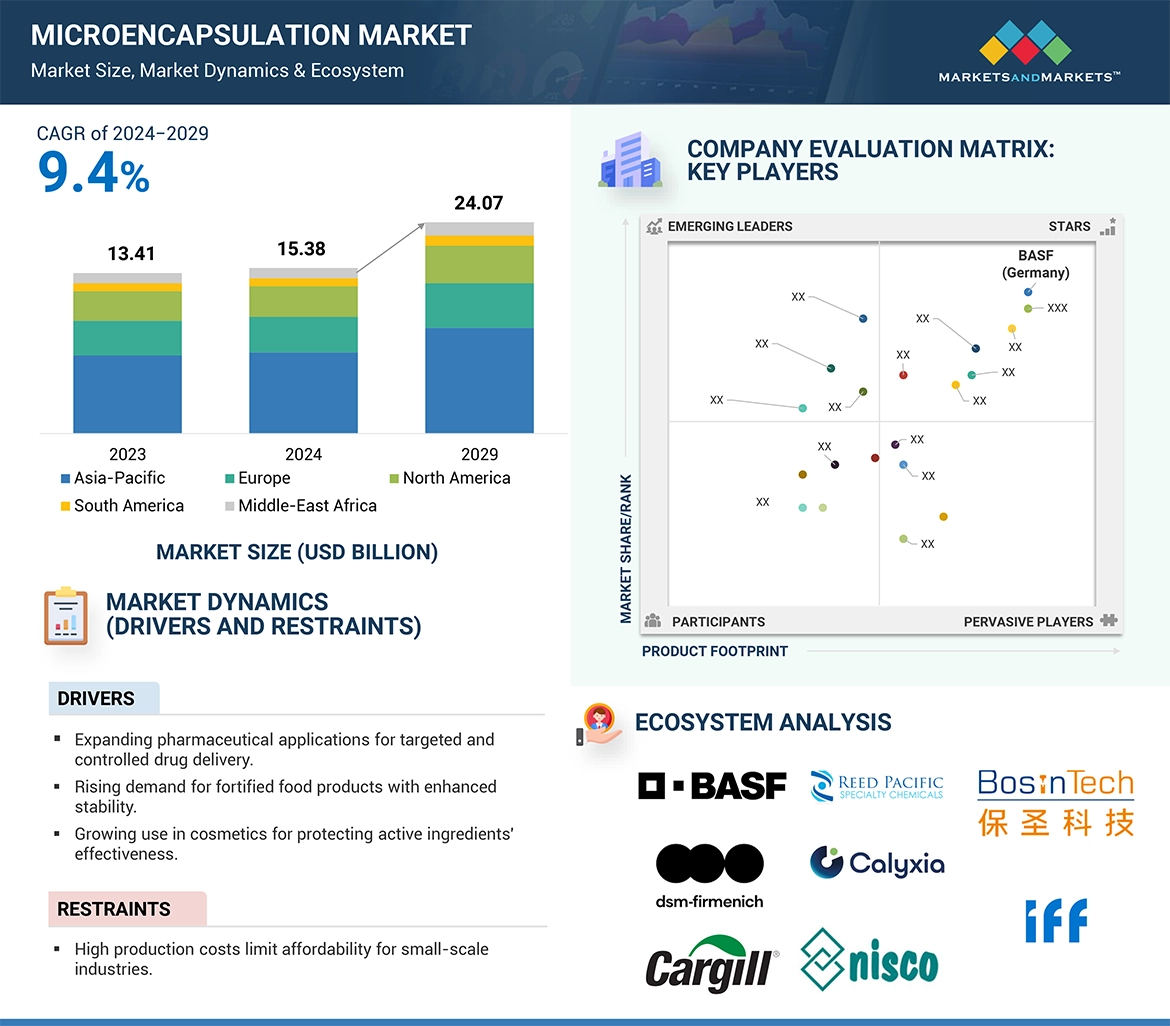
Key Takeaways from the Basal Cell Carcinoma Pipeline Report
- In April 2025, Regeneron Pharmaceuticals announced a phase 2 Study of REGN2810, a Fully Human Monoclonal Antibody to Programmed Death-1, in Patients With Advanced Basal Cell Carcinoma Who Experienced Progression of Disease on Hedgehog Pathway Inhibitor Therapy, or Were Intolerant of Prior Hedgehog Pathway Inhibitor Therapy.
- In April 2025, Replimune Inc. conducted a phase 1B/2 study is a multicenter, open-label, study of RP1 to investigate the (a) objective response rate, in addition to (b) safety and tolerability of RP1 for the treatment of advanced cutaneous malignancies in up to 65 evaluable organ transplant recipients. This will include patients with either previous renal, hepatic, heart, lung, or other solid organ transplantation or hematopoietic cell transplant and experiencing subsequent documented locally advanced or metastatic cutaneous malignancies. The study will enroll a total of 65 evaluable patients.
- In April 2025, Galderma R&D announced a study that two contralateral areas (5×10 cm^2) with skin lesions within the participant were compared. One area was received Metvix PDT at defined intervals and the other was received lesion specific treatment at the discretion of the investigator.
- DelveInsight’s Basal Cell Carcinoma Pipeline report depicts a robust space with 25+ active players working to develop 25+ pipeline therapies for Basal Cell Carcinoma treatment.
- The leading Basal Cell Carcinoma Companies such as PellePharm, MedC Biopharma Corporation, AiViva BioPharma, MediWound, Kintara Therapeutics, IO Biotech, Sirnaomics, Aresus Pharma, Epitome Pharmaceuticals, Transgene, Senhwa Biosciences, Palvella Therapeutics, Suzhou Kintor Pharmaceuticals, Leaf Vertical, and others.
- Promising Basal Cell Carcinoma Therapies such as BO-112, SP-002, Vismodegib, Nivolumab, Ipilimumab, Relatlimab, Vismodegib, and others.
Stay informed about the cutting-edge advancements in Basal Cell Carcinoma treatments. Download for updates and be a part of the revolution in cancer care @ Basal Cell Carcinoma Clinical Trials Assessment
Basal Cell Carcinoma Emerging Drugs
- Patidegib: PellePharm
Patidegib is an investigational small molecule that inhibits hedgehog signaling. It is a topical treatment designed to mitigate the tumor burden in patients with Gorlin Syndrome and Basal Cell Carcinomas (BCCs), and other potential indications. Topical patidegib is a first-in-class topical gel formulation of a proprietary hedgehog inhibitor exclusively licensed from Infinity Pharmaceuticals. Patidegib is currently evaluated in Phase III clinical trials for the treatment of BCCs.
- AIV001: AiViva BioPharma
AIV001 is a novel formulation of a multi-kinase inhibitor combined with AiViva’s proprietary delivery technology, designed for prolonged drug release via intradermal treatment. AIV001 targets multiple pathways to reduce fibroplasia in overlapping phases of wound healing and scarring; targets VEGFR to limit the inflammation and fibrosis associated with rosacea; and inhibits or reduces neovascularization and cell proliferation associated with certain cancers. AIV001 is in Phase I/II clinical studies for the treatment of patients with Basal Cell Carcinoma.
The Basal Cell Carcinoma pipeline report provides insights into
- The report provides detailed insights about companies that are developing therapies for the treatment of Basal Cell Carcinoma with aggregate therapies developed by each company for the same.
- It accesses the Different therapeutic candidates segmented into early-stage, mid-stage, and late-stage of development for Basal Cell Carcinoma Treatment.
- Basal Cell Carcinoma Companies are involved in targeted therapeutics development with respective active and inactive (dormant or discontinued) projects.
- Basal Cell Carcinoma Drugs under development based on the stage of development, route of administration, target receptor, monotherapy or combination therapy, a different mechanism of action, and molecular type.
- Detailed analysis of collaborations (company-company collaborations and company-academia collaborations), licensing agreement and financing details for future advancement of the Basal Cell Carcinoma market.
Learn more about Basal Cell Carcinoma Drugs Opportunities in our groundbreaking Basal Cell Carcinoma Research and development projects @ Basal Cell Carcinoma Unmet Needs
Basal Cell Carcinoma Companies
PellePharm, MedC Biopharma Corporation, AiViva BioPharma, MediWound, Kintara Therapeutics, IO Biotech, Sirnaomics, Aresus Pharma, Epitome Pharmaceuticals, Transgene, Senhwa Biosciences, Palvella Therapeutics, Suzhou Kintor Pharmaceuticals, Leaf Vertical, and others.
Basal Cell Carcinoma pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration
- Oral
- Parenteral
- Intravitreal
- Subretinal
- Topical
- Molecule Type
Basal Cell Carcinoma Products have been categorized under various Molecule types such as
- Monoclonal Antibody
- Peptides
- Polymer
- Small molecule
- Gene therapy
- Product Type
Discover the latest advancements in Basal Cell Carcinoma treatment by visiting our website. Stay informed about how we’re transforming the future of oncology @ Basal Cell Carcinoma Market Drivers and Barriers, and Future Perspectives
Scope of the Basal Cell Carcinoma Pipeline Report
- Coverage- Global
- Basal Cell Carcinoma Companies- PellePharm, MedC Biopharma Corporation, AiViva BioPharma, MediWound, Kintara Therapeutics, IO Biotech, Sirnaomics, Aresus Pharma, Epitome Pharmaceuticals, Transgene, Senhwa Biosciences, Palvella Therapeutics, Suzhou Kintor Pharmaceuticals, Leaf Vertical, and others.
- Basal Cell Carcinoma Therapies- BO-112, SP-002, Vismodegib, Nivolumab, Ipilimumab, Relatlimab, Vismodegib, and others.
- Basal Cell Carcinoma Therapeutic Assessment by Product Type: Mono, Combination, Mono/Combination
- Basal Cell Carcinoma Therapeutic Assessment by Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
For a detailed overview of our latest research findings and future plans, read the full details of Basal Cell Carcinoma Pipeline on our website @ Basal Cell Carcinoma Emerging Drugs and Companies
Table of Content
- Introduction
- Executive Summary
- Basal Cell Carcinoma: Overview
- Pipeline Therapeutics
- Therapeutic Assessment
- Basal Cell Carcinoma – DelveInsight’s Analytical Perspective
- Late Stage Products (Phase III)
- Patidegib: PellePharm
- Drug profiles in the detailed report…..
- Mid Stage Products (Phase II)
- IO103: IO Biotech
- Drug profiles in the detailed report…..
- Early Stage Products (Phase I/II)
- AIV001: AiViva BioPharma
- Drug profiles in the detailed report…..
- Preclinical and Discovery Stage Products
- Cannabidiol: Leaf Vertical
- Drug profiles in the detailed report…..
- Inactive Products
- Basal Cell Carcinoma Key Companies
- Basal Cell Carcinoma Key Products
- Basal Cell Carcinoma- Unmet Needs
- Basal Cell Carcinoma- Market Drivers and Barriers
- Basal Cell Carcinoma- Future Perspectives and Conclusion
- Basal Cell Carcinoma Analyst Views
- Appendix
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Yash Bhardwaj
Email: Send Email
Phone: 09650213330
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/report-store/basal-cell-carcinoma-basal-cell-epithelioma-market
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Basal Cell Carcinoma Clinical Trials and Studies 2025: EMA, PDMA, FDA Approvals, Mechanism of Action, ROA, NDA, IND, and Companies