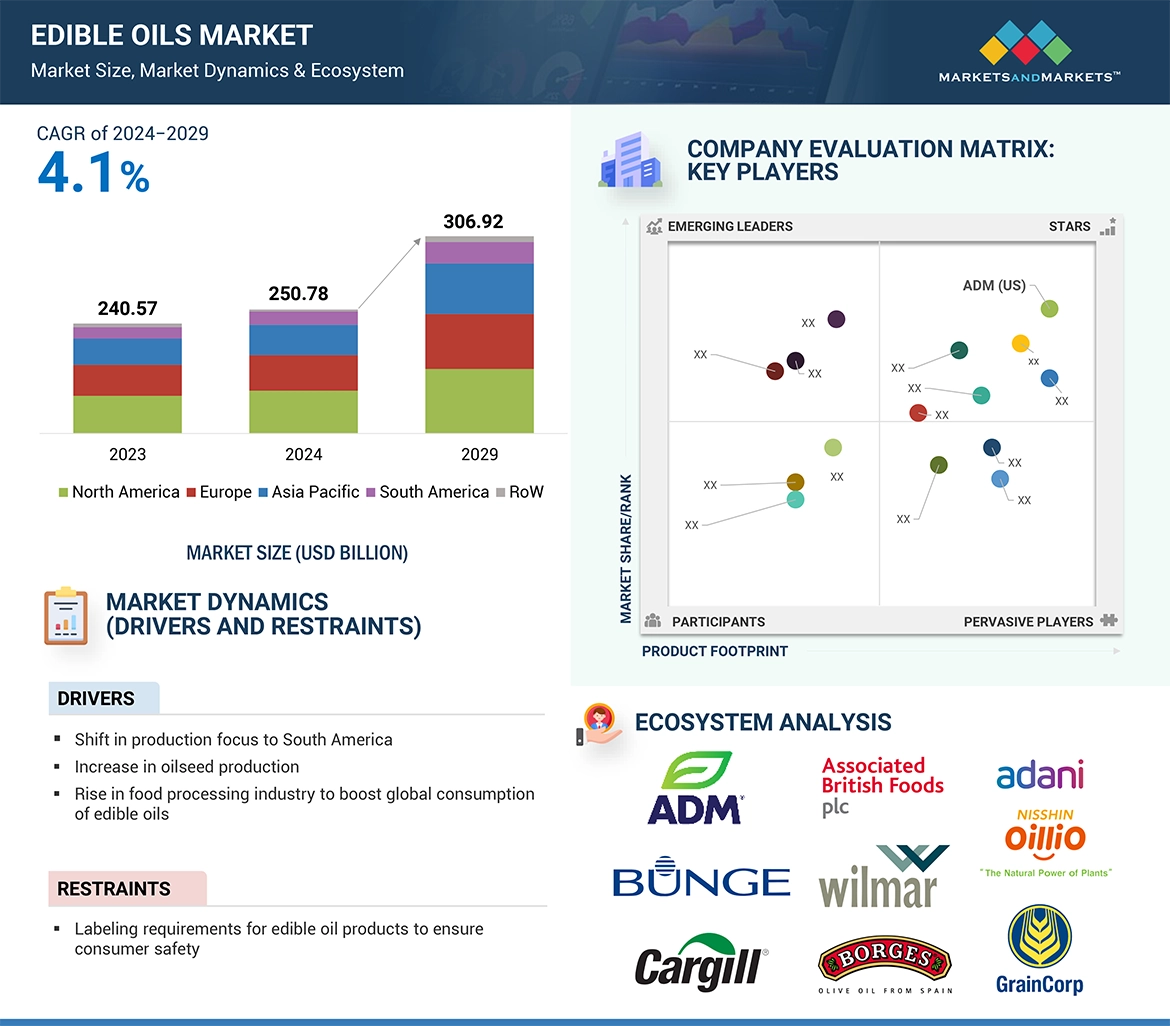
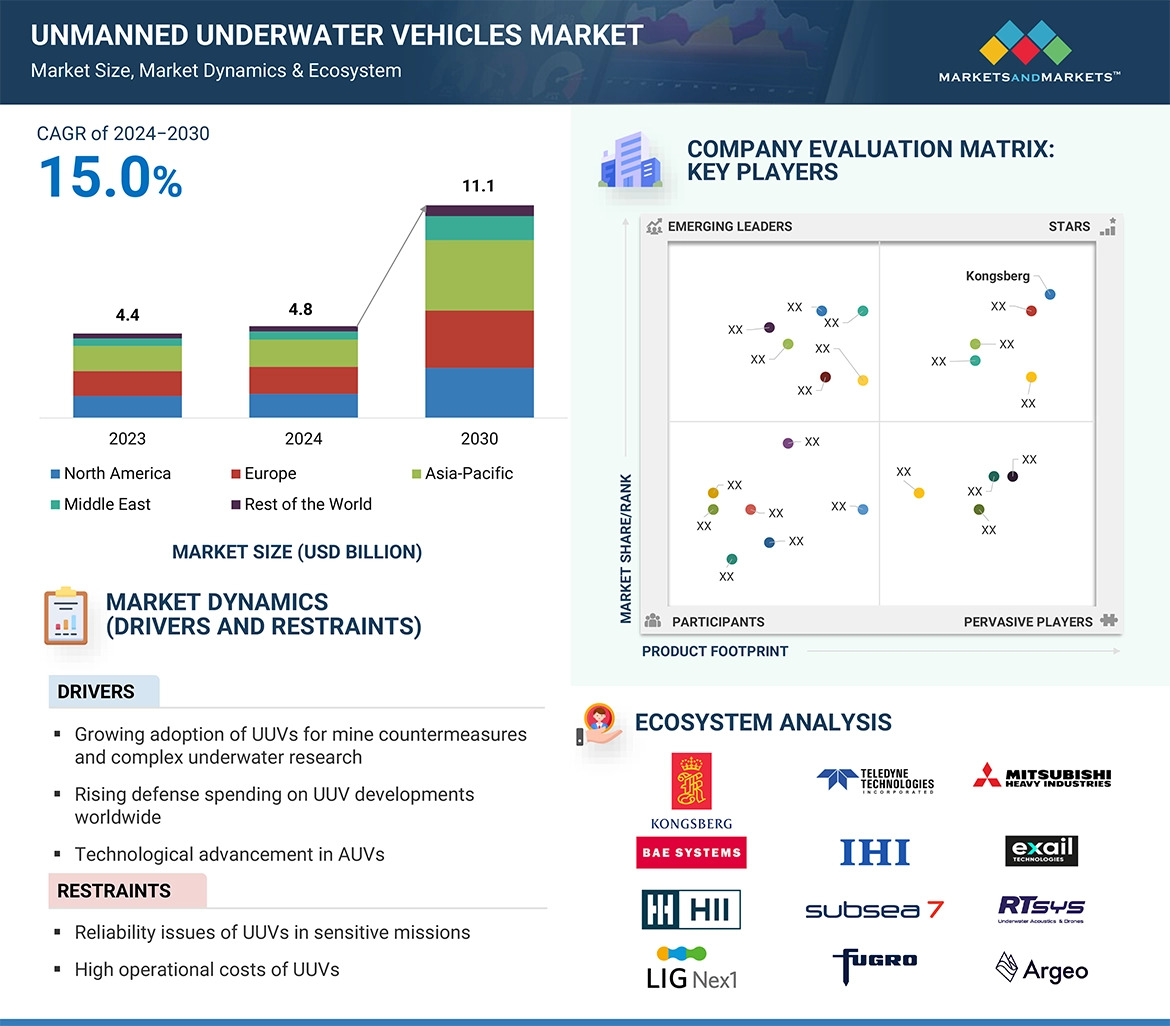
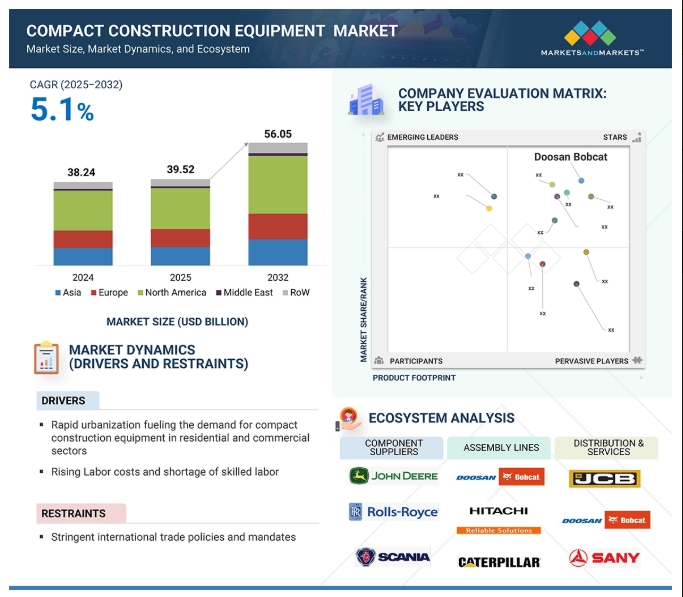
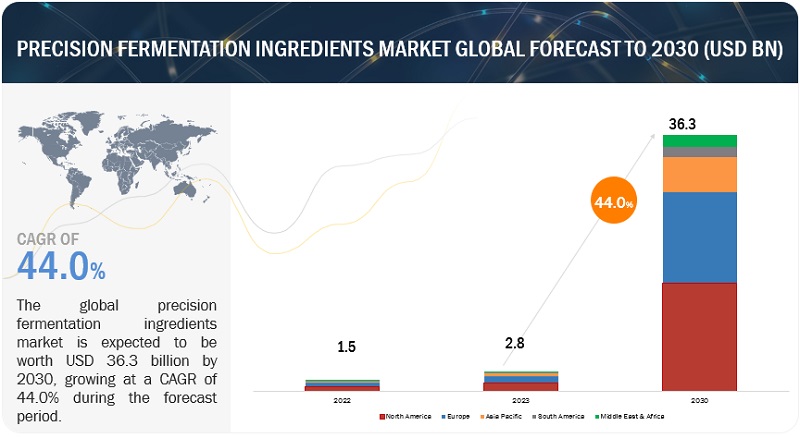
The global precision fermentation ingredients market is projected to grow from USD 2.8 billion in 2023 to USD 36.3 billion by 2030, reflecting a robust CAGR of 44.0% in value. This growth is driven by shifting consumer preferences toward veganism, rising protein consumption, and increased investments in innovation. Advances in genetic engineering have significantly enhanced the efficiency and sustainability of precision fermentation, enabling the reprogramming of microorganisms through synthetic biology to produce specialized food protein components cost-effectively.
Precision Fermentation Ingredients Market Growth Drivers
The growth of the Precision Fermentation Ingredients Market is driven by several key factors:
- Rising Demand for Sustainable and Ethical Food Products: Consumers are increasingly concerned with sustainability, animal welfare, and environmental impacts. Precision fermentation offers a sustainable method to produce ingredients like proteins, flavors, and enzymes without relying on animal-based sources, driving demand in plant-based foods and other eco-friendly alternatives.
- Advancements in Biotechnology and Fermentation Techniques: Continuous improvements in fermentation technology, such as more efficient microbial strains and enhanced fermentation processes, are reducing production costs and increasing the scalability of precision fermentation, making it an attractive option for ingredient production.
- Growth of the Plant-based Food Industry: As plant-based food products gain popularity, there is a need for precision fermentation to create high-quality ingredients like proteins and enzymes that can be used to replicate the taste, texture, and nutritional profile of animal-derived products.
- Rising Demand for Alternative Proteins: The global shift towards plant-based diets and the search for sustainable protein sources is propelling the demand for precision fermentation-derived proteins like those found in meat alternatives, dairy-free products, and other functional foods.
- Regulatory Support for Biotech Innovations: Governments around the world are providing funding and favorable regulations for the development of innovative food production methods, including precision fermentation. This regulatory environment helps to reduce barriers to market entry and drives investment in the sector.
- Consumer Preferences for Healthier and Clean Label Products: The growing preference for clean label foods with fewer artificial additives and more natural ingredients is benefiting precision fermentation, as it can provide ingredients that align with this trend.
- Increased Investment from Major Corporations: Large food and beverage companies are investing heavily in precision fermentation to expand their product portfolios and gain a competitive edge in the rapidly growing market for alternative ingredients and food solutions.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=30824914
Cosmetics & Others Segment to Lead with the Highest CAGR
The cosmetics & others segment is set for remarkable growth, projected to achieve the highest CAGR in the precision fermentation ingredients market. This surge aligns with the 2022 cosmetic trend, emphasizing the long-term benefits of fermented products. These formulations enhance skin absorption by breaking down substances, allowing vital nutrients to penetrate more effectively than non-fermented alternatives. A key player in this space, Geltor specializes in fermented ingredients for hair and skincare, further driving the segment’s expansion.
Meat & Seafood Segment to Dominate in Food & Beverage Applications
The meat & seafood segment is expected to witness the highest CAGR within the food & beverages precision fermentation ingredients market. This innovative approach leverages microorganisms like yeast, bacteria, fungi, and algae to produce plant-based proteins and fats, creating meat alternatives that replicate traditional textures, juiciness, and nutrition. The rising consumer preference for sustainable, health-conscious food choices is fueling the growth of this segment, reflecting the market’s shift towards environmentally friendly and nutritious options.
Request Sample pages: https://www.marketsandmarkets.com/requestsampleNew.asp?id=30824914
Heme Protein Segment to Experience Unprecedented Growth
Heme protein is projected to be the fastest-growing segment in the precision fermentation ingredients market, driven by advancements in microbial fermentation. This process primarily uses genetically modified yeast strains, such as Saccharomyces cerevisiae, to produce heme proteins in a sustainable and efficient manner, eliminating reliance on animal-derived sources. The United States remains a hub for research and development in this field, particularly in heme production for plant-based meat alternatives. Industry leaders like MycoTechnology Inc. and Impossible Foods are at the forefront of this innovation, showcasing the growing demand for fermentation-driven solutions across the alternative protein landscape.
The Asia Pacific region is dominate the precision fermentation ingredients market share.
This growth is driven by rapid urbanization and shifting consumer lifestyles, which have fueled demand for specialized products like alternative proteins and sustainable ingredients—key areas where precision fermentation excels. The region’s strong emphasis on technological advancements and innovation further supports the expansion of this market, creating a dynamic ecosystem for research and development. Additionally, consumers across Asia Pacific are increasingly open to adopting innovative products, demonstrating not just curiosity but active purchasing intent. Furthermore, rising investments and business expansions in the region are contributing to the market’s accelerated growth.
Top 10 Companies in the Precision Fermentation Ingredients Market
- Geltor (US)
- Perfect Day, Inc. (US)
- The Every Co. (US)
- Impossible Foods Inc. (US)
- Motif FoodWorks, Inc. (US)
- Imagindairy Ltd. (Israel)
- Shiru, Inc. (US)
- Formo (Germany)
- Eden Brew (Australia)
- Change Foods (US)
Geltor is a prominent bio-design firm that pioneers in advanced designer proteins. Integrating fermentation, biology, and protein optimization, Geltor produces sustainable and efficient alternative proteins with enhanced functionality. Their 100% animal-free and GMO-free ingredients are globally scalable and cultivated sustainably. Leveraging microbes, the company employs a fermentation process to create products using plant-based and renewable resources. In 2023, Geltor introduced Nucoll and Caviance, novel products catering to haircare and skincare needs. This evolution signifies a transformative approach to beauty and wellness, harnessing biotechnology’s potential for creating effective and eco-friendly alternatives.
Perfect Day, Inc. stands out as a prominent manufacturer of animal-free dairy products. It employs safe and established fermentation techniques to create animal-free whey protein using microflora. In contrast to conventional whey production methods, Perfect Day’s whey protein powder production achieves remarkable environmental efficiencies: a 99% reduction in water usage, a 97% decrease in greenhouse gas emissions, and approximately 60% lower non-renewable energy consumption. These figures are verified through ISO-compliant, third-party-reviewed life cycle assessments (LCAs). The company has established partnerships with multiple brands, expanding its presence in over 5,000 stores across the United States. Its market influence extends across North America and the Asia Pacific region as well.
Impossible Foods is engaged in providing plant-based fish, dairy, and meat products. The company’s product portfolio includes various plant-based products such as burgers, sausages, chicken nuggets, pork, and meatballs. Impossible Foods includes soy leghemoglobin manufactured through precision fermentation into its plant-based burger to provide the end product with the desired meat-like texture and taste. The company has its manufacturing facility located in Oakland, CA, US. Additionally, the company has partnered with one of the foremost food processing companies in the US, OSI Group, which provides extra manufacturing space to produce the popular Impossible Sausage and Impossible Burger. As a globally recognized entity, Impossible Foods serves customers across multiple countries, including the United States, Australia, Canada, Hong Kong, Macau, New Zealand, Singapore, the United Arab Emirates, and the United Kingdom.
Precision Fermentation Ingredients Industry News:
In May 2023, The EVERY Company and Alpha Foods officially entered into a Joint Development Agreement with the shared objective of bringing next-generation alt-meat products to the market. This collaboration aims to leverage EVERY’s trailblazing expertise in animal-free protein production and Alpha Foods’ renowned chef-crafted plant-based foods to accelerate advancements in taste and texture for non-animal products.
In March 2023, the partnership between The Hartman Group, Perfect Day, and Cargill enabled Perfect Day to gain valuable insights into consumer attitudes and preferences related to precision fermentation ingredients. This data has helped Perfect Day strengthen its position in the market, spearhead the Precision fermentation Alliance
In October 2022, Impossible Foods’ collaboration with Domino’s Australia, introducing Impossible™ Beef on their pizzas, strategically expands their market presence, boosts brand exposure, and appeals to environmentally conscious consumers.
About MarketsandMarkets™
MarketsandMarkets™ has been recognized as one of America’s Best Management Consulting Firms by Forbes, as per their recent report.
MarketsandMarkets™ is a blue ocean alternative in growth consulting and program management, leveraging a man-machine offering to drive supernormal growth for progressive organizations in the B2B space. With the widest lens on emerging technologies, we are proficient in co-creating supernormal growth for clients across the globe.
Today, 80% of Fortune 2000 companies rely on MarketsandMarkets, and 90 of the top 100 companies in each sector trust us to accelerate their revenue growth. With a global clientele of over 13,000 organizations, we help businesses thrive in a disruptive ecosystem.
The B2B economy is witnessing the emergence of $25 trillion in new revenue streams that are replacing existing ones within this decade. We work with clients on growth programs, helping them monetize this $25 trillion opportunity through our service lines – TAM Expansion, Go-to-Market (GTM) Strategy to Execution, Market Share Gain, Account Enablement, and Thought Leadership Marketing.
Built on the ‘GIVE Growth’ principle, we collaborate with several Forbes Global 2000 B2B companies to keep them future-ready. Our insights and strategies are powered by industry experts, cutting-edge AI, and our Market Intelligence Cloud, KnowledgeStore™, which integrates research and provides ecosystem-wide visibility into revenue shifts.
In addition, MarketsandMarkets SalesIQ enables sales teams to identify high-priority accounts and uncover hidden opportunities, helping them build more pipeline and win more deals with precision.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com/Market-Reports/precision-fermentation-market-30824914.html
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Precision Fermentation Ingredients Market to Witness Rapid Growth: Industry Trends and Future Projections