Baby Safety Seats Industry Overview
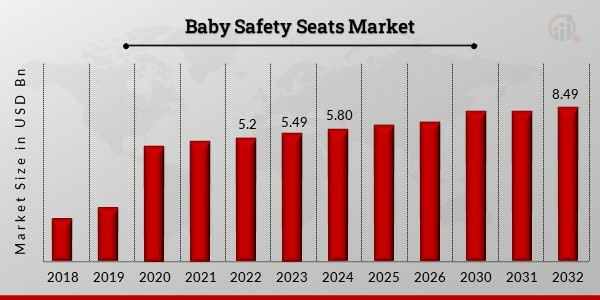
In 2023, Baby Safety Seats Market Size was estimated to be worth USD 5.49 billion. The market for baby safety seats is expected to expand at a compound annual growth rate (CAGR) of 4.88% from 2024 to 2032, from USD 5.80 billion in 2023 to USD 8.49 billion by 2032. The main factors driving market growth are rising public awareness of road safety, strict government regulations, and growing parental concerns about their children’s safety.
The global baby safety seats market has experienced a steady expansion, driven by factors such as rising birth rates, growing urbanization, and increased disposable income. In recent years, parents have become more conscious of the risks associated with unrestrained children in vehicles, leading to higher adoption rates of baby safety seats. Additionally, advancements in design and technology have made these seats more effective, user-friendly, and comfortable for infants and toddlers.
“Free Sample Copy” – Access a complimentary copy of our report to explore its content and insights
https://www.marketresearchfuture.com/sample_request/8117
Competitive Landscape
The baby safety seats market is highly competitive, with key players focusing on innovation, product quality, and compliance with safety regulations.
Some of the prominent companies in the industry include:
- Artsana Group Bellelli S.r.l.
- Britax Child Safety Inc.
- Combi Corporation
- Diono LLC
- Dorel Juvenile Group
- Goodbaby International Holdings Ltd.
- Graco Children Products Inc.
- Jane Group
- Kiwi Baby
- Newell Brands Inc.
- Nuna International BV
- RECARO Holding GmbH
- Thule Group
- TOPEAK INC.
These companies continuously invest in research and development to enhance product efficiency, durability, and safety. Collaborations with regulatory authorities and participation in awareness campaigns also help in strengthening their market presence.
Key Market Drivers
- Stringent Safety Regulations and Laws – Governments in several countries have mandated the use of child restraint systems, making it legally necessary for parents to invest in high-quality safety seats. For example, in the U.S., the National Highway Traffic Safety Administration (NHTSA) enforces strict child safety seat laws to reduce fatalities and injuries.
- Rising Consumer Awareness – Parents are now more informed about the potential dangers of traveling without a baby safety seat. Campaigns and educational programs by organizations like the World Health Organization (WHO) and Safe Kids Worldwide have significantly contributed to increased consumer awareness.
- Technological Advancements – The integration of smart features such as temperature control, impact resistance, and enhanced harness systems has improved the safety and comfort of baby safety seats. Leading brands are investing in research and development to introduce innovative products that cater to different consumer needs.
- E-Commerce Growth – The expansion of online retail platforms has made baby safety seats more accessible to consumers. With the convenience of home delivery, customer reviews, and a wide range of options, more parents are opting to purchase safety seats through e-commerce channels.
Market Challenges
Despite the promising growth, the baby safety seats market faces some challenges:
- High Costs – Premium baby safety seats with advanced safety features can be expensive, limiting their adoption in price-sensitive markets.
- Counterfeit Products – The presence of low-quality, counterfeit products poses a significant risk to child safety, leading to concerns about product authenticity.
- Lack of Awareness in Developing Regions – In certain regions, particularly in developing countries, the adoption of baby safety seats remains low due to limited awareness and the absence of strict regulations.
Market Segmentation
The baby safety seats market can be segmented based on product type, distribution channel, and region:
- By Product Type:
- Infant Car Seats (0-12 months)
- Convertible Car Seats (Birth-4 years)
- Booster Seats (4+ years)
- Combination Seats
- By Distribution Channel:
- Online Retail
- Specialty Stores
- Supermarkets & Hypermarkets
- Others
- By Region:
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
“Proceed to Buy” – Move forward with your purchase and gain instant access to the complete report
https://www.marketresearchfuture.com/checkout?currency=one_user-USD&report_id=8117
Regional Insights
North America leads the global market, primarily due to stringent safety regulations and high consumer awareness. The U.S. and Canada have strict child restraint laws, and manufacturers continuously innovate to meet safety standards.
Europe follows closely, with countries such as Germany, France, and the UK emphasizing road safety for children. The European Union’s R129 (i-Size) regulation has contributed to market growth by setting new safety standards.
Asia-Pacific is expected to witness the highest growth rate in the coming years. Rising disposable income, increasing birth rates, and growing awareness in countries like China, India, and Japan are major driving factors.
Future Trends and Opportunities
The baby safety seats market is expected to grow at a steady pace due to continuous advancements and increasing consumer demand. Some key trends shaping the future of this market include:
- Smart Baby Safety Seats: The integration of IoT-enabled features such as sensors, mobile app connectivity, and automatic alerts will enhance safety measures.
- Sustainable and Eco-Friendly Materials: With growing environmental concerns, manufacturers are focusing on producing eco-friendly baby car seats using recyclable and non-toxic materials.
- Enhanced Safety Standards: Regulatory bodies are expected to introduce more stringent safety measures, ensuring that manufacturers comply with global safety standards.
- Growth in Emerging Markets: Increasing urbanization and improved economic conditions in developing nations will create new opportunities for market expansion.
Discover more Research Reports on Consumer-and-Retail Industry, by Market Research Future:
Feminine Hygiene Market Research Report Forecast Till 2032
Smokeless Tobacco Market Research Report Forecast Till 2030
Hair Care Market Research Report Forecast Till 2032
Eyewear Market Research Report Forecast Till 2032
Cosmetics Market Research Report Forecast Till 2032
About Market Research Future:
Market Research Future (MRFR) is a global market research company that takes pride in its services, offering a complete and accurate analysis with regard to diverse markets and consumers worldwide. Market Research Future has the distinguished objective of providing optimal quality research and granular research to clients. Our market research studies by products, services, technologies, applications, end users, and market players for global, regional, and country level market segments, enable our clients to see more, know more, and do more, which help answer your most important questions.
Contact Us:
Market Research Future (Part of Wantstats Research and Media Private Limited)
99 Hudson Street, 5Th Floor New York, 10013
United States of America
+1 628 258 0071 (US)
+44 2035 002 764 (UK)
Email: info@marketresearchfuture.com
Website: https://www.marketresearchfuture.com/
https://www.wiseguyreports.com/
Media Contact
Company Name: Market Research Future
Contact Person: Media Relations
Email: Send Email
Country: United States
Website: https://www.marketresearchfuture.com
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Baby Safety Seats Market is Predicted to Register A CAGR Of 4.88% and Reach USD 8.49 Billion By 2032