The Idiopathic Membranous Nephropathy market report also offers comprehensive insights into the Idiopathic Membranous Nephropathy market size, share, Idiopathic Membranous Nephropathy epidemiology, emerging therapies, market drivers and barriers, ongoing clinical trials, key collaboration in the space, market uptake by key therapies and companies actively pushing Idiopathic Membranous Nephropathy market size growth forward.
Some of the key highlights from the Idiopathic Membranous Nephropathy Market Insights Report:
-
Several key pharmaceutical companies, including Bristol Myers Squibb Company, Merck and Co Inc., Mylan Pharmaceutical ltd., Novartis AG, Pfizer Inc., Roche, Aspen Global Inc., Astellas Pharma Inc., Baxter Healthcare Corporation, Sigma Aldrich Corporation, and others, are developing novel products to improve the Idiopathic Membranous Nephropathy treatment outlook.
-
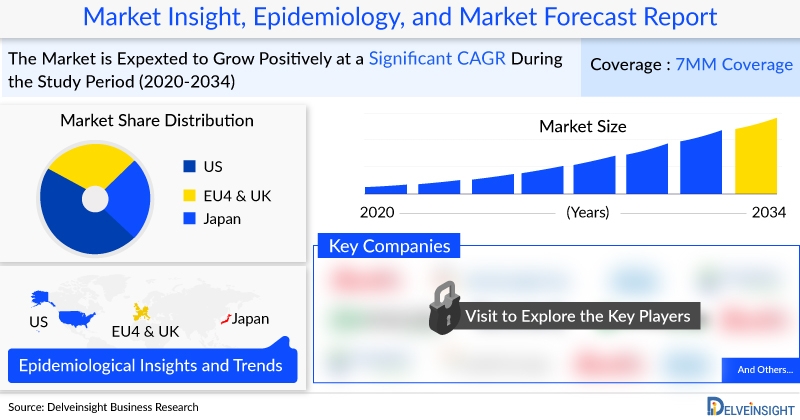
The Idiopathic Membranous Nephropathy (IMN) market in the 7MM is projected to reach approximately USD 1,000 million by 2034. In 2023, the United States held the largest market share, generating around USD 88 million, surpassing EU4 (Germany, Spain, Italy, France), the United Kingdom, and Japan.
-
IMN is a major glomerular disease characterized by excessive protein loss in the urine (nephrotic syndrome) and is primarily autoimmune in nature. Currently, there are no approved treatments specifically for IMN, with management focused on supportive care. However, the market is expected to grow due to improved uptake of existing drugs, upcoming therapy launches, and increased awareness.
-
In 2023, the estimated total prevalent IMN population in the 7MM was approximately 71,000 cases. The US accounted for about 40% of these cases, while EU4 and the UK collectively made up around 35%, and Japan contributed approximately 25%, making it the second-largest region after the US.
-
Several emerging therapies are in development, including TNT119, ALPN-303, MOR202, SNP-ACTH (1-39) Gel, and GAZYVA. Notably, in October 2023, the FDA granted Breakthrough Therapy designation (BTD) to felzartamab for primary membranous nephropathy. Additionally, in June 2023, the FDA awarded orphan drug designation (ODD) to SNP-ACTH (1-39) gel for treating primary membranous nephropathy.
-
In July 2024, Biogen finalized its acquisition of Human Immunology Biosciences (HI-Bio). Earlier, in June 2022, HI-Bio had entered an agreement with MorphoSys AG to develop and commercialize the anti-CD38 antibody felzartamab.
-
As per DelveInsight analysis, the Idiopathic Membranous Nephropathy market is anticipated to witness growth at a considerable CAGR
Strategise your business goals by understanding market dynamics @ Idiopathic Membranous Nephropathy Market Landscape
Idiopathic Membranous Nephropathy Overview
Idiopathic Membranous Nephropathy is a common glomerular disease closely linked to nephrotic syndrome. It occurs when the immune system mistakenly attacks the glomeruli, resulting in excessive protein loss in urine (proteinuria), swelling (edema), and, in some cases, progression to kidney failure. The term “idiopathic” indicates that no clear underlying cause has been identified, though the condition may also develop due to autoimmune disorders or infections. This disease primarily affects adults, particularly middle-aged men, and manifests through symptoms such as foamy urine, leg and abdominal swelling, and weight gain caused by fluid retention.
Diagnosis involves a combination of clinical evaluation and laboratory tests. Urine analysis detects proteinuria, often exceeding 3.5 g/day, while blood tests assess kidney function, low albumin levels (hypoalbuminemia), and lipid imbalances (dyslipidemia), which are typical of nephrotic syndrome. A kidney biopsy remains the gold standard for diagnosis, revealing immune complex deposits along the glomerular capillary walls. Additionally, serological testing for antibodies like anti-PLA2R helps distinguish primary Idiopathic Membranous Nephropathy from secondary forms, aiding in precise treatment decisions.
Do you know the treatment paradigms for different countries? Download our Idiopathic Membranous Nephropathy Market Sample Report
Idiopathic Membranous Nephropathy Epidemiology Insights
-
In 2023, the United States represented approximately 40% of the total prevalent cases of Idiopathic Membranous Nephropathy across the 7MM.
-
Meanwhile, Japan’s prevalence data indicates a relatively steady trend projected over the forecast period from 2024 to 2034.
Idiopathic Membranous Nephropathy Epidemiology Segmentation
DelveInsight’s Idiopathic Membranous Nephropathy market report is prepared on the basis of epidemiology model. It offers comprehensive insights to the Idiopathic Membranous Nephropathy historical patient pools and forecasted Idiopathic Membranous Nephropathy patients. The report provides in-depth data of various subtypes and for the same epidemiology is segmented further. The Idiopathic Membranous Nephropathy Market report proffers epidemiological analysis for the study period 2020-34 in the 7MM segmented into:
-
Idiopathic Membranous Nephropathy Prevalence
-
Age-Specific Idiopathic Membranous Nephropathy Prevalence
-
Gender-Specific Idiopathic Membranous Nephropathy Prevalence
-
Diagnosed and Treatable Cases of Idiopathic Membranous Nephropathy
Visit for more @ Idiopathic Membranous Nephropathy Epidemiological Insights
Idiopathic Membranous Nephropathy Market Outlook
Key companies, including Hoffmann-La Roche, HI-Bio, Cerium Pharmaceuticals, BeiGene, and others, are advancing their lead candidates through various stages of clinical development. Their goal is to explore these products as treatments for Idiopathic Membranous Nephropathy.
In the 7MM, MOR202 (felzartamab) is projected to achieve the highest market revenue, reaching approximately USD 500 million by 2034. Among the EU4 and the UK, Germany had the largest market share, with an estimated value of around USD 10 million in 2023. Japan’s market is anticipated to grow to approximately USD 70 million by 2034. In 2023, Rituximab generated roughly USD 50 million in revenue across the 7MM.
Idiopathic Membranous Nephropathy Emerging Drugs
-
GAZYVA (obinutuzumab): Hoffmann-La Roche
-
SNP-ACTH (1-39) Gel: Cerium Pharmaceuticals
Idiopathic Membranous Nephropathy Key Companies
-
Bristol Myers Squibb Company, Merck and Co Inc., Mylan Pharmaceutical ltd., Novartis AG, Pfizer Inc., Roche, Aspen Global Inc., Astellas Pharma Inc., Baxter Healthcare Corporation, Sigma Aldrich Corporation, and others
For more information, visit Idiopathic Membranous Nephropathy Market Analysis, Patient Pool, and Emerging Therapies
Scope of the Idiopathic Membranous Nephropathy Market Report:
-
11 Years Forecast
-
7MM Coverage
-
Descriptive overview of Idiopathic Membranous Nephropathy, causes, signs and symptoms, diagnosis, treatment
-
Comprehensive insight into Idiopathic Membranous Nephropathy epidemiology in the 7MM
-
Idiopathic Membranous Nephropathy marketed and emerging therapies
-
Idiopathic Membranous Nephropathy companies
-
Idiopathic Membranous Nephropathy market drivers and barriers
Table of Contents:
1 Idiopathic Membranous Nephropathy Market Key Comprehensive Insights
2 Idiopathic Membranous Nephropathy Market Report Introduction
3 Competitive Intelligence Analysis for Idiopathic Membranous Nephropathy
4 Idiopathic Membranous Nephropathy Market Analysis Overview at a Glance
5 Executive Summary of Idiopathic Membranous Nephropathy
6 Idiopathic Membranous Nephropathy Epidemiology and Market Methodology
7 Idiopathic Membranous Nephropathy Epidemiology and Patient Population
8 Idiopathic Membranous Nephropathy Patient Journey
9 Idiopathic Membranous Nephropathy Treatment Algorithm, Idiopathic Membranous Nephropathy Current Treatment, and Medical Practices
10 Key Endpoints in Idiopathic Membranous Nephropathy Clinical Trials
11 Idiopathic Membranous Nephropathy Marketed Therapies
12 Idiopathic Membranous Nephropathy Emerging Therapies
13 Idiopathic Membranous Nephropathy: 7 Major Market Analysis
14 Attribute analysis
15 Access and Reimbursement Overview of Idiopathic Membranous Nephropathy
16 Idiopathic Membranous Nephropathy Market Key Opinion Leaders Reviews
18 Idiopathic Membranous Nephropathy Market Drivers
19 Idiopathic Membranous Nephropathy Market Barriers
20 SWOT Analysis
21 Disclaimer
22 DelveInsight Capabilities
23 About DelveInsight
Related Reports:
Idiopathic Membranous Nephropathy Epidemiology 2034
DelveInsight’s “Idiopathic Membranous Nephropathy – Epidemiology Forecast to 2034” report delivers an in-depth understanding of the disease, historical and forecasted Idiopathic Membranous Nephropathy epidemiology in the 7MM, i.e., the United States, EU5 (Germany, Spain, Italy, France, and the United Kingdom), and Japan.
Idiopathic Membranous Nephropathy Pipeline 2024
“Idiopathic Membranous Nephropathy Pipeline Insights, 2024” report by DelveInsight outlines comprehensive insights of present clinical development scenarios and growth prospects across the Idiopathic Membranous Nephropathy market. A detailed picture of the Idiopathic Membranous Nephropathy pipeline landscape is provided, which includes the disease overview and Idiopathic Membranous Nephropathy treatment guidelines.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Kritika Rehani
Email: Send Email
Phone: +14699457679
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: Nevada
Country: United States
Website: https://www.delveinsight.com/
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Idiopathic Membranous Nephropathy Market, 2034 | Bristol Myers Squibb Company, Merck and Co Inc., Mylan Pharmaceutical ltd., Novartis AG, Pfizer Inc., Roche, Aspen Global Inc., Astell