Frost & Sullivan released a report, titled “Healthnice: Building an Integrated, End-to-End R&D and Manufacturing Service Platform with Innovative R&D and High-end Preparation Technologies.” The report delves into the development trends of high-end preparations and highlights how Healthnice, by closely following industry development trends and precisely positioning itself in the field of high-end preparations and modified new drugs, continuously enhances its competitiveness in the pharmaceutical industry. This is achieved through the establishment of a dual-track business model, integrated full-industry-chain service capabilities, a quality-first approach, and a strategy of deep domestic roots with an international outlook, thereby providing high-quality and efficient R&D and production services to global clients.
I. Aligning with Industry Trends: Healthnice Advances Step by Step
Amid the global pharmaceutical industry’s evolution, high-end preparations have become a mainstream trend in drug development. In 2016, China’s Ministry of Industry and Information Technology identified high-end preparations as a key development area, recognized for their superior compliance, safety, and convenience. This category includes novel injectable delivery systems like liposomes and nanoformulations, as well as immediate-release oral dosage forms and transdermal drug delivery systems.
High-end preparations are the preferred choice for upgrading and innovating pharmaceutical products, providing patients with improved therapeutic options. At the same time, as centralized procurement of drugs becomes normalized and national healthcare cost-control measures tighten, generic drugs are entering an era of slim profit margins. Low-value drugs with significant product homogeneity face a higher risk of market elimination. Therefore, for pharmaceutical companies, high-end preparations represent a high-value, cost-effective strategy for differentiation. By strategically investing in key products within the high-end preparation sector, companies can enhance product value and extend lifecycle, thereby building core competitiveness. The substantial commercial potential and market impact of high-end preparations have attracted more companies to invest in this field. To address the escalating market competition, strategies such as outsourcing R&D and manufacturing services or directly acquiring technological achievements are becoming essential for improving R&D quality and accelerating development timelines.
Healthnice has recognized these industry trends, building momentum and gradually evolving into a comprehensive CXO pharmaceutical company with independent project development at its core. The company specializes in high-end preparations and modified new drugs, focusing on high-value and high-potential products while actively exploring new opportunities. Through sustained technological expertise and R&D investments, Healthnice has established a fully integrated, end-to-end R&D and manufacturing system. This closed-loop industry chain spans innovative drug and high-end preparation development, custom manufacturing, and commercial production. Empowering the pharmaceutical industry with high-quality services, Healthnice has provided approximately 700 multi-mode, end-to-end R&D and manufacturing solutions to approximately 300 clients worldwide.
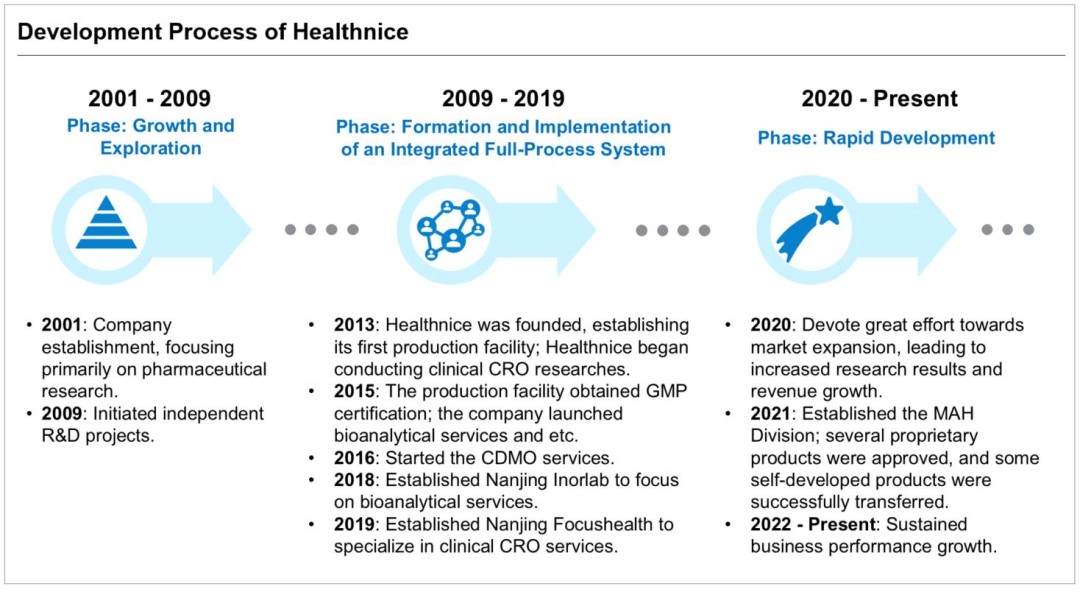
Healthnice’s development journey showcases its innovative capabilities and stands as a microcosm of the pharmaceutical industry’s transformation. Starting with early exploration in generic drug pharmaceutical research in 2001, the company pivoted toward a specialization transformation in 2007, following the introduction of the Measures for the Administration of Drug Registration (No.28). Keeping pace with industry trends, Healthnice transitioned from a single quality research service model to a strategic, diversified business model, systematically developing parallel services alongside quality research. Through strategic product selection and forward-looking project planning, Healthnice has successfully established a comprehensive pharmaceutical research system, built a professional R&D team, and accumulated a rich portfolio of projects.
Over the past decade, Healthnice has developed a fully integrated service chain. In 2009, the company initiated its independent R&D projects. Guided by a scientific and forward-looking product selection system, Healthnice has focused on unmet clinical needs, identifying high-value products and launching independent R&D projects. In 2016, the company formally established its CDMO business segment. With the integration of critical functions such as production, clinical research, and bioanalysis services, Healthnice gradually shaped its comprehensive service chain, reaching a mature development stage in 2019.
Healthnice has strategically focused on high-end preparations and modified new drugs, intensifying its market expansion efforts since 2020. The successful approval of several proprietary products and the transfer of marketing authorizations for self-developed products have significantly enhanced the company’s efficiency in translating research into practice and boosted its revenue growth. Looking ahead, as market dynamics shift toward greater consolidation and regulatory requirements for R&D capabilities and compliance become more stringent, Healthnice is aligning its growth with these industry trends. The company is actively initiating and undertaking more projects in modified new drugs with high technical content and added value. These efforts aim to upgrade its business model, further strengthen its competitive advantages, and solidify its position in the pharmaceutical industry.
II. Integrated, End-to-End Full Industry Chain Services Provide High-Quality, Efficient R&D and Manufacturing Services to Clients
Dual-Track Model: A Unique Model to Unlock New Expansion Opportunities
Healthnice has implemented a dual-track business model that integrates parallel pathways: entrusted R&D services and the transfer of self-developed projects, further enhanced by an equity-sharing mechanism to fully maximize and elevate project value. Under this model, the company achieves a win-win relationship with its clients. For clients, Healthnice’s proprietary project transfer services ensure a high-quality R&D process, effectively enhancing research efficiency and accelerating new drug launches. Moreover, clients can entrust their selected projects to Healthnice, benefiting from its full-chain, one-stop high quality services from R&D to production and its extensive development expertise to achieve “ready-to-market” convenience. For Healthnice, the dual-track approach lays the foundation for a diversified business model. On one hand, entrusted R&D services provide project certainty, establishing a stable revenue stream and client base. On the other hand, converting R&D achievements enhances the company’s pricing power, profitability, and innovation capabilities.
Currently, Healthnice’s business operations are on a positive growth trajectory, supported by a dynamic and tiered R&D pipeline of “developing, commercializing, and reserving” projects. Leveraging years of pharmaceutical R&D experience and sharp market insights, the company has secured approvals for 50 projects over the past two years. As of the first half of 2024, Healthnice has 336 projects in its pipeline, including 95 proprietary projects retained as company assets and 241 projects in transfer-stage development. Since 2022, the company has initiated more than 100 new projects annually, maintaining a well-structured progression across its pipeline. This sustained growth ensures a consistent flow of high-quality products entering the market, continuously enriching the company’s product portfolio and providing robust support for future business expansion.
Service Chain Innovation: Building a Fully Integrated, End-to-End Industry Chain
Building on extensive experience in the pharmaceutical industry, Healthnice has developed a seamless and integrated business model focused on pharmaceutical research and development over more than a decade of growth. The year 2013 marked a significant milestone: the company began constructing its pharmaceutical production facility and established a clinical research team, marking its full-scale entry into the pharmaceutical services sector. In 2015, Healthnice successfully obtained GMP certification, laying a solid foundation for preparation production and quality management. In 2016, the company gradually expanded into CDMO services, providing comprehensive support from research to production. By 2018, Healthnice established its subsidiary, Nanjing Inorlab, dedicated to bioanalysis services, followed by the establishment of Nanjing Focushealth in 2019, which specializes in independently managing clinical research.
In recent years, the increasing demand for pharmaceutical R&D outsourcing has created unprecedented opportunities for the CRO industry. In this content, many CRO companies have pursued acquisitions, partnerships, and other strategies to enhance their service capabilities. While the industry races toward integrated and seamless models, Healthnice has prioritized business innovation from its early stages. Through the close collaboration of Institutes of Biomedical Research and three subsidiaries, the company has actively explored new development models and directions, achieving a fully integrated pharmaceutical R&D and production service chain.
Today, Healthnice operates under an “Integrated tripartite model” comprising pharmaceutical research, clinical studies, and manufacturing. This model breaks down the traditional barriers supplier-segmented drug development stages, significantly reducing communication costs and preventing information bottlenecks. By enabling seamless integration across all stages and parallel processing of multiple steps, Healthnice’s end-to-end, integrated service model offers clients more efficient, convenient, and comprehensive pharmaceutical R&D and production solutions.
Driving Innovation: Strengthening R&D in high-end preparations and Expanding Competitive Product Portfolios
Healthnice has established a multidimensional, comprehensive technology platform matrix encompassing “APIs + preparations” and “R&D + clinical + manufacturing.” The company now operates five key technology platforms: preparation R&D, API R&D, high-end preparation technology transfer and validation, clinical research, and bioanalytical testing. These platforms are supported by 11 core technologies, including polymer coating-based taste-masking microsphere delivery systems and solid dispersion solubilization techniques. Additionally, the company has established 14 drug delivery platforms, covering various dosage forms such as cataplasms and blow-fill-seal (BFS) eye drops.
As drug development enters the era of preparation innovation, high-end preparations have become the strategic focus for pharmaceutical companies. However, R&D for high-end preparations involves high technical barriers, complex manufacturing processes, and stringent quality control requirements. Broadly, the main challenges in this process can be summarized into three key aspects: First, maintaining strict quality control of excipients remains a significant challenge. Second, high-end preparations demand highly stringent control over process parameters. Finally, the complexity of bioequivalence evaluation for high-end preparations has substantially increased. An industrialization platform that integrates solutions to these challenges is critical for achieving independent innovation. Supported by its 5 key platforms and 11 core technologies, Healthnice has established an innovative preparation industrialization research platform capable of addressing complex technical issues during product development. This platform provides comprehensive support to both generic and innovative pharmaceutical companies entering the high-end preparation sector. For instance, within its clinical trial platform, the bioequivalence technology platform for special dosage forms has successfully overcome challenges in trial design and process management. By accumulating extensive experience in trial design and project management, the company has significantly improved the success rate and efficiency of clinical trials.
Healthnice fully leverages the strengths of its innovative preparation research platform to focus on developing high-value self-developed drugs. Within the high-end preparation technology sector, the company strategically targets unmet clinical needs as the foundation for developing novel preparation technologies. It has made strategic deployments in areas such as sustained and controlled release, transdermal drug delivery, inhalation therapies, ophthalmic applications, and peptide-based. Currently, the company is undertaking the development of a series of products, including Diclofenac Epolamine Patch, Lidocaine Cataplasms, and Ketoprofen Cataplasms.
Among Healthnice’s successfully commercialized R&D outcomes are several self-developed products that are the first or only of their kind in China. For instance, the Tenofovir disoproxil fumarate, utilizing polymer membrane coating technology via coacervation method, became the first generic drug of its kind in granule form to gain regulatory approval in China. This formulation effectively masks the taste without compromising drug release, improving patient compliance. Another notable product is the Omeprazole and Sodium Bicarbonate, used to treat acid-related diseases such as peptic ulcers. It is the first and only oral proton pump inhibitor (PPI) approved for critically ill patients. Despite the original drugs not being approved in China for years, Healthnice successfully developed omeprazole and sodium bicarbonate dry suspension (I) and (II), which received approval in 2021, making them the first of their kind in the domestic market. In the field of transdermal drug delivery, the company has established a proprietary technology platform and extensive R&D experience, leading to a growing number of contracted R&D projects and successful product commercialization. Moreover, the company places great focuses on meeting clinical needs for ophthalmic drugs. Since 2020, it has expanded it development into eye drops category, including Cyclosporin eye drops (III), Tafluprosttrade eye drops, Sodium hyaluronate eye drops, and Brimonidine tartrate eye drops.
Building on its innovative R&D capabilities and expertise in high-end preparation technology, the company has independently initiated ten modified new drug projects, primarily focusing on the gastrointestinal and anti-inflammatory/pain relief therapies. Six of these projects have successfully completed technology translation. Looking ahead, Healthnice will continue to address clinical needs, further explore the potential of modified drugs, and continuously expand its innovative drug development pipeline to deliver greater benefits to patients.
Quality as Priority: Building a Production System That Balances Compliance and Scalability, Enhancing the Quality of Back-End Manufacturing Services
Healthnice has transformed its extensive R&D and production experience into a solid foundation for expanding its manufacturing services. By leveraging the collaborative strengths of its subsidiaries: Nanjing Inorlab, Nanjing Focushealth, and Healthnice Pharmaceutical, the company has strategically extended its business into the CDMO sector. This expansion enables Healthnice to offer clients value-added technical R&D services and production capacity support. At the same time, the extension into CDMO has enabled Healthnice to form a complete value chain of drug R&D, custom manufacturing, and commercial production, enhancing its competitiveness and profitability.
The company’s production system integrates efficient manufacturing with comprehensive risk management. In terms of regulation compliance, Healthnice adheres to regulatory standards from China, the US, and Europe, while actively advancing digital transformation. By adopting advanced online management systems, it ensures real-time monitoring of the entire production process, provides instant tracking of contract manufacturing projects, and dynamically manages potential risks. These measures greatly improves project management efficiency and improve flexibility in mitigating potential risks. Moreover, Healthnice continues to expand and strengthen its quality control team, further broadening and deepening the scope of its quality management system.
Healthnice is also advancing its production capacity to enhance service resilience. Under careful planning for both approved and pipeline products, the company initiated Phase II base construction expansion to mitigate the risks associated with product co-production. This project takes approximately 12,000 square meters and includes facilities and workshops for oral solutions and oral solid dosage forms. The expansion will enable a significant capacity increase, with granules/suspensions (calculated at 6g/bag) soaring from 50 million bags to 500 million bags annually. Similarly, capacity of oral solution (calculated at 100mL/bottle) will increase from 6 million bottles to 30 million bottles annually. Additionally, the company has prioritized operational efficiency and cost control, reserving 50% of its designed capacity to flexibly accommodate future approved products and anticipated market demand growth.
III. Deepening Roots Domestically While Expanding Globally
Healthnice strengthens its industry competitiveness through a balanced focus on both domestic and global growth. Domestically, the company continues to strengthen its core business and expand service offerings. Globally, it is committed to providing comprehensive, one-stop services for the entire lifecycle of high-end preparations and innovative drugs, covering both R&D and manufacturing.
“Focusing on Refinement”: Aiming to Lead in High-End Transdermal Preparations
Healthnice aims to deepen its presence in high-end preparation fields such as sustained and controlled release, transdermal, inhalable, ophthalmic, and peptide-based formulations. In the field of transdermal patch, its portfolio includes cataplasms, hot-melt adhesives tapes, and solventborne adhesive patches. Since 2019, the company has proactively invested in building production facilities for cataplasms, successfully developing several products while gaining extensive manufacturing expertise and technical knowledge.
Healthnice plans to solidify its leadership in the transdermal patch field in the coming future, by increasing R&D investment in hot-melt adhesive tapes and solventborne adhesive technologies. Simultaneously, it will plan and implement new production lines to ensure efficient, high-quality responses to market demands.
“Layering Foundations and Achieving New Growth”: Advancing the Industrialization of APIs and Preparations
To address challenges in cost control, stable supply of APIs, and controlled sourcing of key excipients, Healthnice is actively strengthening its R&D efforts for APIs and its excipients. By adopting a flexible collaboration model, the company invests in early-stage R&D and partners with high-quality API manufacturers for subsequent production and registration, ensuring a stable supply of APIs and maintaining cost advantages. Additionally, Healthnice is working to establish its own API production base to enhance its industrial capabilities.
The company is also exploring the field of highly potent drugs, building specialized production facilities to provide integrated R&D and manufacturing services. Looking ahead, Healthnice plans to undertake commissioned R&D and production of highly potent drugs, creating new business growth opportunities.
Global Expansion: Strategic International Market Development
The internationalization of preparations is crucial for upgrading China’s API industry and optimizing its export structure. Developing high-end preparations represents a critical step for China’s pharmaceutical sector in global expansion. Healthnice adopts a phased expansion strategy, advancing from APIs to preparations. The company initially targets markets in Southeast Asia, South America, and Africa with APIs, establishing a strong foundation for future expansion into the U.S. and European markets. Meanwhile, the company actively engages in the international CDMO business for advanced intermediates, further expanding its global market presence. Building on its success in API expansion, Healthnice is dedicated to securing FDA and EMA certifications for its preparation production facilities and product lines. This will enable the company to launch high-technology, differentiated preparations in overseas markets through registration and approval processes. Ultimately, Healthnice aims to establish an integrated strategy covering key intermediates, APIs, and high-end preparations, achieving full coverage of the pharmaceutical value chain.
In addition, Healthnice actively seeks strategic partnerships with foreign or multinational pharmaceutical companies (MNCs). By offering comprehensive CXO services, including preclinical research, clinical trials, registration, and contract manufacturing, the company supports the entry of overseas drugs into the Chinese market. These collaborations not only facilitate market access for foreign clients but also provide strong support for their growth in China, acting as the connection bridge to the Chinese pharmaceutical market.
The full content can also be viewed on the Frost & Sullivan global official website: https://www.frost.com/news/press-releases/building-an-integrated-end-to-end-rd-and-manufacturing-service-platform-with-innovative-rd-and-high-end-preparation-technologies/
Media Contact
Company Name: Frost & Sullivan
Contact Person: Qian Li
Email: Send Email
Country: China
Website: http://www.frostchina.com