The report “Biocompatible Coatings Market by Type (Antibacterial, hydrophilic), Material (Polymer, Ceramics, Metal), End-Use Industry (Healthcare, Food & Beverage, Medical Devices), & Region (APAC, Europe, North America, MEA & SA) – Global Forecast to 2028”, size is estimated at USD 14.6 billion in 2022 to USD 29.7 billion by 2028, at a CAGR of 12.4%, between 2023 and 2028. The market research report provides access to critical information such as biocompatible coatings market growth drivers, market growth restraints, current market trends, the market’s economic and financial structure, and other key market details. The rapid growth of the healthcare industry, Rapid urbanization, population growth, and the increasing focus on hygiene and cleanliness drive the biocompatible coatings market. The biocompatible coatings market is divided into five key regions:Asia Pacific, Europe, North America, Europe, the Middle East & Africa, and South America. China and India, among other countries, are fastest growing biocompatible coatings market globally.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=32624649
The demand for biocompatible coatings is influenced by several factors. One crucial factor is the increasing focus on healthcare and medical advancements. With the continuous progress in medical technology and the growing emphasis on patient well-being, there is a higher demand for biocompatible coatings in various applications. These coatings play a vital role in enhancing the biocompatibility of medical devices, implants, and prosthetics, reducing the risk of adverse reactions and improving patient outcomes. The aging population and the prevalence of chronic diseases drive the demand for medical devices and implants, which, in turn, fuels the demand for biocompatible coatings. The companies have adopted various strategies, such as mergers & acquisitions, investment & expansion, joint ventures, & new product launches and to increase their global presence and maintain sustained growth in the biocompatible coatings market.
Polymer segment is expected to account for the largest share in 2022.
Polymers provide versatility, allowing for the development of coatings with tailored properties such as biocompatibility, durability, flexibility, and resistance to wear and corrosion. These characteristics make polymer coatings suitable for a diverse range of medical devices and implants, including orthopedic implants, cardiovascular stents, surgical instruments, and drug delivery systems. advancements in polymer technology have enabled the development of biodegradable coatings. These coatings gradually dissolve or degrade over time, reducing the need for additional surgical interventions to remove the coating. Bioresorbable polymer coatings are particularly advantageous for implantable devices, as they can facilitate tissue healing and integration. The widespread adoption of polymer-based biocompatible coatings can be attributed to their compatibility with various fabrication techniques, cost-effectiveness, and well-established manufacturing processes. These factors make polymer coatings more accessible and easier to integrate into existing medical device production workflows.
Get a Sample Copy of This Report: https://www.marketsandmarkets.com/requestsampleNew.asp?id=32624649
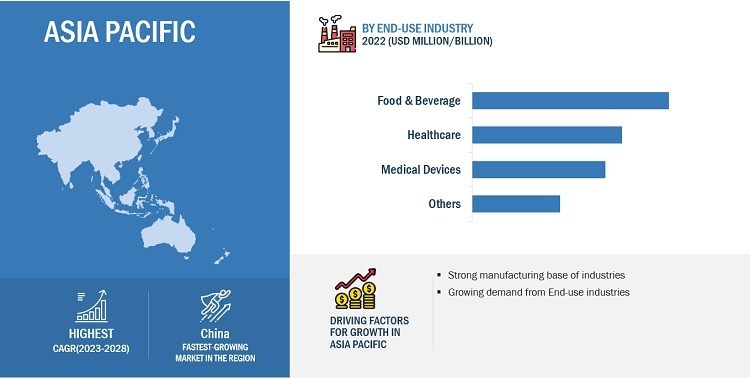
Food & Beverage segment is expected to account for the largest share in 2022.
The food and beverage segment is a crucial component of the biocompatible coatings industry, and it plays a vital role in ensuring the safety, quality, and shelf life of food and beverage products. Food and beverage packaging serves as a protective barrier that safeguards products from external factors such as moisture, light, air, and contaminants. It helps prevent spoilage, maintain product freshness, and extend shelf life. Advanced packaging materials and technologies, including biocompatible coatings, are employed to ensure optimal product protection and preservation. the food and beverage packaging segment is driven by the need for product protection, safety, branding, convenience, sustainability, and regulatory compliance. Biocompatible coatings play a significant role in enhancing the safety, functionality, and aesthetics of packaging materials in this segment, meeting the evolving demands of both consumers and regulatory bodies.
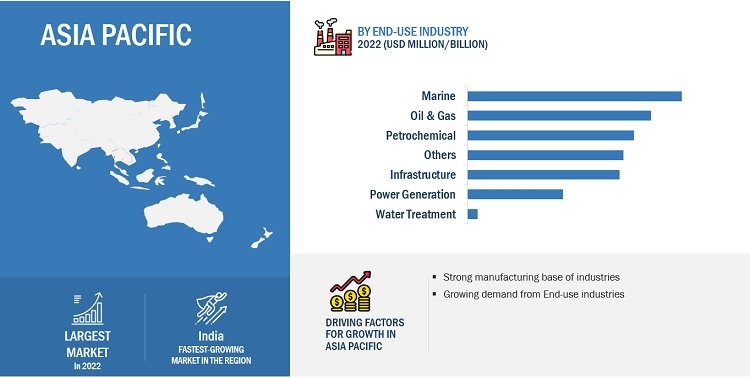
Asia Pacific is expected to register highest CAGR during the forecast period.
Asia Pacific region has a large and rapidly growing population, along with increasing healthcare expenditure. This demographic trend drives the demand for medical devices, implants, and related technologies, which in turn fuels the need for biocompatible coatings. The region’s rising middle class and improving healthcare infrastructure also contribute to the market growth.
There is a growing focus on medical tourism in countries like India, Thailand, and Malaysia, where patients from other countries seek medical treatments. This trend further drives the demand for advanced medical devices and implants, which often require biocompatible coatings. the region’s manufacturing capabilities and cost advantages attract global medical device manufacturers to establish production facilities in Asia Pacific countries. This factor supports the local production and consumption of biocompatible coatings, further contributing to market growth.
Biocompatible Coatings Market Companies
The major players in the global biocompatible coatings market are SurModics, Inc. (US), DSM Biomedical (US), Hydromer Inc. (US), Covalon Technologies Ltd. (Canada), and Hemoteq AG (Germany). These players opted for inorganic growth through mergers & acquisitions, agreements, and partnerships. For instance, in July 2021, SurModics, Inc. has acquired privately-held Vetex Medical Limited. The Galway, Ireland-based medical device developer and manufacturer has focused exclusively on venous clot removal solutions.
SurModics, Inc: SurModics, Inc. is a global provider of surface modification and in vitro diagnostic technologies. The company is headquartered in Eden Prairie, Minnesota, United States. SurModics has been operating for over four decades and has established itself as a leader in the field of biocompatible coatings and surface modification technologies. The company’s core focus is on developing and commercializing innovative solutions that enhance the performance, biocompatibility, and functionality of medical devices. SurModics offers a broad portfolio of biocompatible coatings designed to address various applications and industries, including cardiovascular, ophthalmology, neurology, and in-vitro diagnostics. SurModics’ biocompatible coatings provide functionalities such as lubricity, hydrophilicity, drug delivery, anti-thrombogenicity, and bioactive capabilities. These coatings are tailored to meet the specific requirements of medical devices, ensuring improved performance, reduced complications, and enhanced patient outcomes. SurModics collaborates with medical device manufacturers, pharmaceutical companies, and diagnostic testing laboratories to develop customized coating and surface modification solutions. The company’s team of scientists and engineers work closely with customers to understand their specific needs and deliver innovative solutions that address their challenges.
Inquire Before Buying: https://www.marketsandmarkets.com/Enquiry_Before_BuyingNew.asp?id=32624649
In April 2020, Cook Medical and Surmodics, Inc. have announced a new distribution agreement in which Cook Medical will market two novel Surmodics products, hydrophilic percutaneous transluminal angioplasty (PTA) balloon catheters that may be utilised beyond 0.014-inch and 0.018-inch wire guides.
DSM Biomedical: DSM Biomedical is a division of DSM, a global science-based company headquartered in the Netherlands. DSM Biomedical specializes in developing and manufacturing biomedical materials and technologies, with a focus on providing innovative solutions for the medical device and healthcare industries. The company offers a wide range of products and solutions that address various medical applications, including orthopedics, cardiovascular devices, dental applications, drug delivery systems, and wound care. DSM Biomedical’s expertise lies in developing biocompatible materials, coatings, drug delivery systems, and regenerative solutions that enhance the performance, biocompatibility, and safety of medical devices. The company places a strong emphasis on sustainability and environmental responsibility. DSM Biomedical strives to develop materials and technologies that are eco-friendly and have minimal impact on the environment. This commitment aligns with the growing demand for sustainable solutions in the medical industry.
About MarketsandMarkets™
MarketsandMarkets™ has been recognized as one of America’s best management consulting firms by Forbes, as per their recent report.
MarketsandMarkets™ is a blue ocean alternative in growth consulting and program management, leveraging a man-machine offering to drive supernormal growth for progressive organizations in the B2B space. We have the widest lens on emerging technologies, making us proficient in co-creating supernormal growth for clients.
Earlier this year, we made a formal transformation into one of America’s best management consulting firms as per a survey conducted by Forbes.
The B2B economy is witnessing the emergence of $25 trillion of new revenue streams that are substituting existing revenue streams in this decade alone. We work with clients on growth programs, helping them monetize this $25 trillion opportunity through our service lines – TAM Expansion, Go-to-Market (GTM) Strategy to Execution, Market Share Gain, Account Enablement, and Thought Leadership Marketing.
Built on the ‘GIVE Growth’ principle, we work with several Forbes Global 2000 B2B companies – helping them stay relevant in a disruptive ecosystem. Our insights and strategies are molded by our industry experts, cutting-edge AI-powered Market Intelligence Cloud, and years of research. The KnowledgeStore™ (our Market Intelligence Cloud) integrates our research, facilitates an analysis of interconnections through a set of applications, helping clients look at the entire ecosystem and understand the revenue shifts happening in their industry.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com/Market-Reports/biocompatible-coating-market-32624649.html
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Biocompatible Coatings Market, Size, Analytical Overview, CAGR, Growth Factors, Demand and Trends Forecast to 2028