Microencapsulation Market by Shell Material (Polymers, Gums & Resins, Lipids, Carbohydrates, and Proteins), Technology, Core Material, Core Material Form, End-Use Industry, Functionality, and Region – Global Forecast to 2029
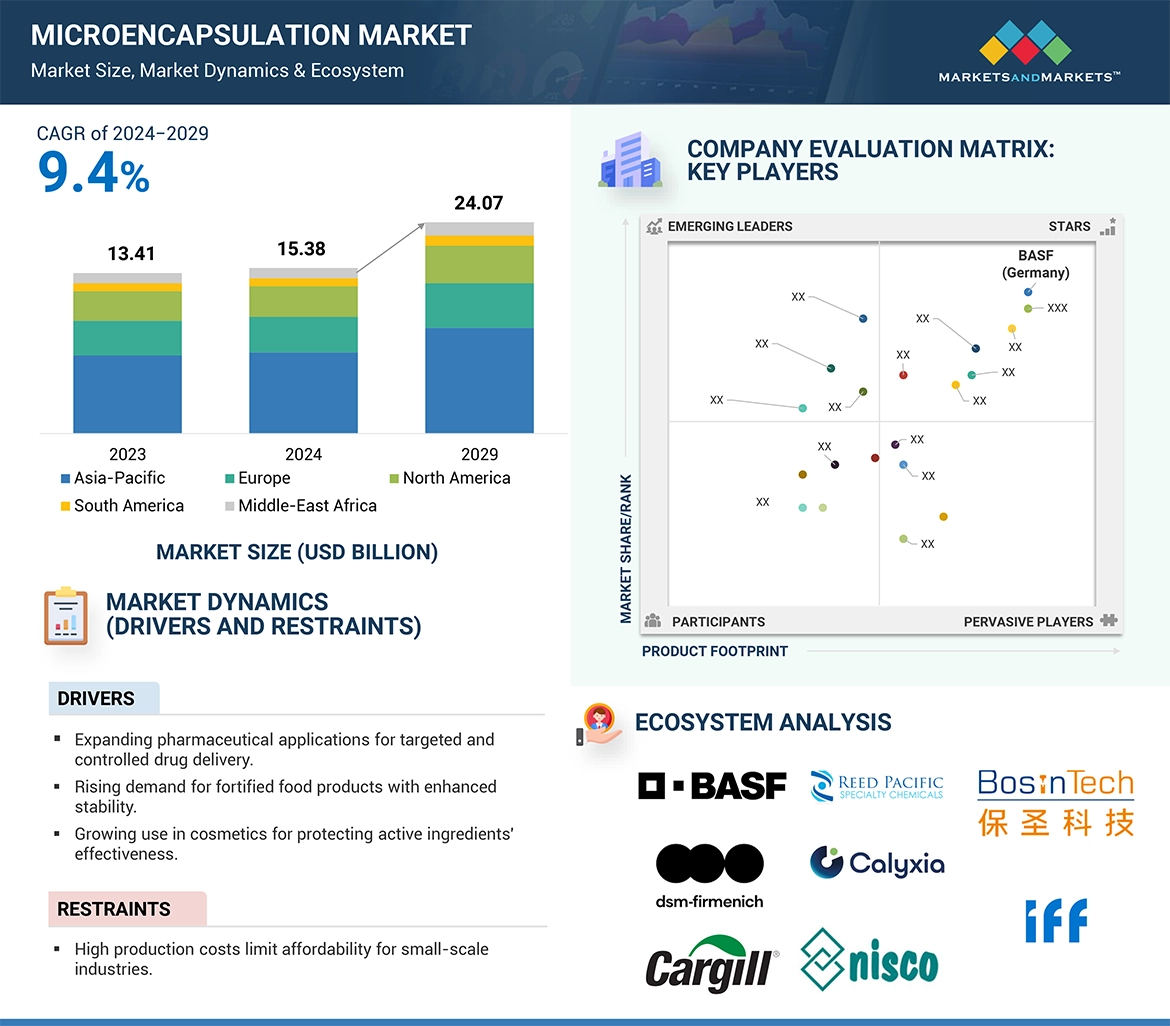
The global microencapsulation market is estimated at USD 15.38 billion in 2024 and is projected to reach USD 24.07 billion by 2029, at a CAGR of 9.4% from 2024 to 2029. An increase in applications of microencapsulation is due to its potential industries across pharmaceutical, food, and personal care products. For instance, within the pharmaceutical industry, controlled drug delivery will streamline with improved effectiveness on treatment outcomes since active ingredients will now be released at specific locations and times within the body, averting side effects while improving patient compliance. Protecting sensitive ingredients such as vitamins, minerals, and probiotics from degradation, thus preserving stability and efficacy till the food is consumed, microencapsulation in the food industry shows an important aspect concerning fortified and functional foods that show an increasing demand these days due to health consciousness among consumers. Improved efficiency and lower production costs resulted from advancements in microencapsulation technologies, such as spray drying and coacervation, making the technology more accessible. This increased the demand for innovative, sustainable, and high-quality products across these industries, pushing the growth of the global microencapsulation market.
Microencapsulation Market Drivers: Demand for Functional Foods
Health-conscious consumers are seeking more than just basic nutrition from food products; the integration of these essential nutrients, probiotics, vitamins, and minerals within food products using microencapsulation technology is particularly enhancing their functional properties. For instance, omega-3 fatty acid-enriched products are gaining recognition because more and more health-conscious consumers are seeking these oils as they are usually drawn from fish oil. Microencapsulation protects the oil from oxidation and degradation and thus preserves its efficacy while increasing shelf life. In May 2024, dsm-firmenich (Switzerland) announced the launch of Humiome® B2, the world’s first ‘biotic’ vitamin, at Vitafoods Europe 2024. Utilizing patented Microbiome Targeted Technology™, this microencapsulation approach ensures targeted delivery of vitamin B2 to the colon, enhancing bioavailability and supporting gut health by nourishing beneficial microbiota, emphasizing holistic health.
This has the additional advantage of being in line with the clean-label trend, wherein consumers prefer fewer additives and preservatives. Microencapsulation thus masks undesirable tastes or odors accompanying certain ingredients, making it possible for manufacturers to include beneficial components without compromising flavor. With the functional food market in full bloom and driven by changing consumer preferences, microencapsulation technology is likely to take center stage in product development and innovation.
Microencapsulation Market Opportunities: Technological Advancements
A new technological approach has been attained with the integration of microencapsulation and artificial intelligence. This technology, along with various biomedical applications, promises a bright future for the collection of industries so involved. Microencapsulation is the enclosure of cells or the active constituent within a polymeric semi-permeable membrane, which allows for controlled release and protects cells and constituents from environmental stimuli. The importance of this method goes a long way in medicine; particularly in regenerative medicine, for it has enabled the improvement in stem cell viability and functionality, tissue repair, and controlled drug delivery.
Incorporation of AI in microencapsulation processes will be efficient in optimizing the process while having precision. The learning algorithms of machines can be used to sense and differentiate microcapsules during manufacture, thereby exercising quality control by segregating desirable capsules from waste materials. This enables doubling the production as well as enhancing yield. Further, AI will model and predict the optimal encapsulation conditions, such as polymer selection and environmental parameters, resulting in better output on the basis of cell viability.
Extensive datasets analyzed from experiments of microencapsulation can help in the identification of patterns and correlations usually overlooked by traditional methods. The synergy that may come from microencapsulation and AI would help revolutionize the field of regenerative medicine as well as drug delivery systems that enable therapies microencapsulated to be far more effective while addressing complex medical challenges.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=83597438
The food and beverage segment are the fastest-growing end user in the microencapsulation market.
Advancements in demand for convenience foods by consumers have promoted manufacturers to create microencapsulation that improves the stability and shelf life of ingredients such as vitamins and probiotics. For instance, probiotic-protected yogurt is a product whose probiotics are microencapsulated so that they become effective until they are consumed.
In addition, the demands placed upon microencapsulation by increasing requirements for functional foods having added benefits beyond plain nutrition help to dictate its use. Such examples include fortified beverages and snack bars, which utilize microencapsulated vitamins or minerals to maximize their health benefit beyond basic needs for immunity or energy. For example, In January 2023, BASF SE (Germany) developed a unique microencapsulation method for vitamins A and D, used in flour and sugar fortification. This process not only extends shelf life but also improves flowability and reduces dust during production, making it highly advantageous for food producers.
The technological improvements in microencapsulation techniques enhance further scalability for companies. For example, DuPont (US) and others are developing novel techniques to control the release of flavors and nutrients in food and beverages, thus enhancing its appeal in the market. The segment in the microencapsulation market, with respect to health, convenience, and innovation, is driving this rapid growth.
The Asia Pacific region is anticipated to experience the fastest growth between 2024 and 2029.
The demand in countries like China, India, and Japan, for fortified and functional products from the food and beverage sector is likely to be the driving force behind growth. As health awareness increases and the trend shifts to healthier lifestyles, vitamins, probiotics, and other nutrients call for improved stability and bioavailability through microencapsulation.
More opportunities for microencapsulation technologies emerge in the Asia Pacific as the pharmaceutical and cosmetics industries grow in the region. Companies in these sectors can rely heavily on encapsulation techniques to enhance controlled active ingredients, including drug formulations and skincare formulas. Most importantly, the governments in the region have tightened the policy of local production with incentives and investments in innovation, which attracts major global players to set up manufacturing and R&D facilities in the regions. For instance, In April 2021, BASF SE (Germany) announced the groundbreaking for the third phase of its Innovation Campus Shanghai, investing approximately USD 296.8 million. This expansion includes new R&D facilities and an academic-sounding board to enhance collaboration with top universities, strengthening BASF’s innovation capabilities to meet the needs of growing industries in Asia.
Leading Microencapsulation Manufacturers:
The report profiles key players such as BASF (Germany), FrieslandCampina (Netherlands), dsm-firmenich (Switzerland), Givaudan (Switzerland), International Flavors & Fragrances Inc. (US), Sensient Technologies Corporation (US), Balchem Corp. (New Jersey), Ingredion (US), Kerry Group plc (Ireland), Symrise (Germany), DuPont (US), Cargill Incorporated (US), Syngenta (Switzerland), Lycored (New Jersey), Koehler Group (US).
Request Sample Pages: https://www.marketsandmarkets.com/requestsampleNew.asp?id=83597438
About MarketsandMarkets™
MarketsandMarketsTM has been recognized as one of America’s best management consulting firms by Forbes, as per their recent report.
MarketsandMarkets™ is a blue ocean alternative in growth consulting and program management, leveraging a man-machine offering to drive supernormal growth for progressive organizations in the B2B space. We have the widest lens on emerging technologies, making us proficient in co-creating supernormal growth for clients.
Earlier this year, we made a formal transformation into one of America’s best management consulting firms as per a survey conducted by Forbes.
The B2B economy is witnessing the emergence of $25 trillion of new revenue streams that are substituting existing revenue streams in this decade alone. We work with clients on growth programs, helping them monetize this $25 trillion opportunity through our service lines – TAM Expansion, Go-to-Market (GTM) Strategy to Execution, Market Share Gain, Account Enablement, and Thought Leadership Marketing.
Built on the ‘GIVE Growth’ principle, we work with several Forbes Global 2000 B2B companies – helping them stay relevant in a disruptive ecosystem. Our insights and strategies are molded by our industry experts, cutting-edge AI-powered Market Intelligence Cloud, and years of research. The KnowledgeStore™ (our Market Intelligence Cloud) integrates our research, facilitates an analysis of interconnections through a set of applications, helping clients look at the entire ecosystem and understand the revenue shifts happening in their industry.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com/Market-Reports/microencapsulation-market-83597438.html
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Microencapsulation Market: Latest Innovations and Emerging Opportunities (2024-2029)











