DelveInsight’s “Parkinson’s Disease Market Insights, Epidemiology, and Market Forecast – 2034” report delivers an in-depth understanding of the Parkinson’s disease, historical and forecasted epidemiology as well as the Parkinson’s disease market trends in the United States, EU4 and the UK (Germany, France, Italy, Spain) and the United Kingdom, and Japan.
Unlock key insights into the Parkinson’s Disease Market! Download DelveInsight’s comprehensive report to explore market trends, pipeline analysis, and emerging therapies @ Parkinson’s Disease Market Size
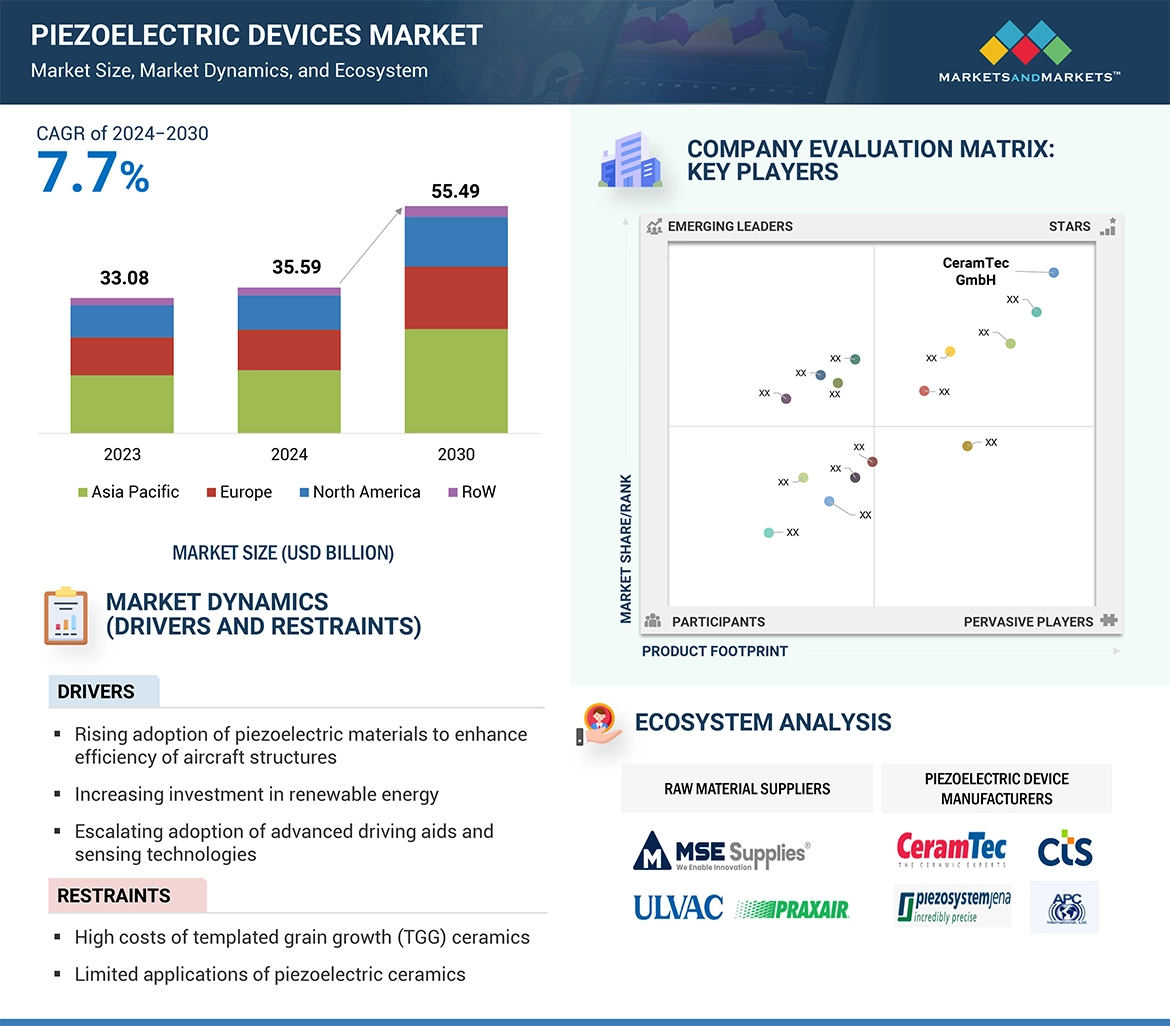
Key Takeaways from the Parkinson’s Disease Market Report
- In January 2025:- UCB Biopharma SRL:- The purpose of the study is to estimate the pharmacodynamic effects of minzasolmin (UCB0599) on brain pathophysiology in Early-start versus Delayed-start participants originally diagnosed with new onset Parkinson’s disease.
- According to DelveInsight’s epidemiology model, the total diagnosed prevalent cases of Parkinson’s disease were approximately 1,210 thousand in the US in 2023 which is expected to increase during the forecast period (2024–2034) due to increasing awareness, aging population, and advancement of diagnostic procedures.
- In the 7MM, Japan accounted for approximately 258 thousand cases diagnosed prevalent cases of Parkinson’s disease in 2023.
- Among the EU4 and the UK, Germany accounted for the highest diagnosed prevalent cases of Parkinson’s disease with nearly 495 thousand cases in 2023 followed by France accounting for approximately 277 thousand cases. On the other hand, Spain accounted for the least with nearly 149 thousand cases.
- In 2023, Japan accounted for approximately 135 thousand cases in males and 123 thousand cases in females, based on gender-specific diagnosed prevalent cases of Parkinson’s disease. These cases are expected to increase during the forecast period.
- In the US, nearly 19 thousand cases were diagnosed with Parkinson’s disease in the age group =49 years, 215 thousand cases in 50–64 years, 449 thousand cases in 65–74 years, and 527 thousand cases in =75 years in 2023.
- Among the stage-specific diagnosed prevalent cases of Parkinson’s disease in Japan, there were approximately 9 thousand cases for Stage I, 40 thousand cases for Stage II, 107 thousand cases for Stage III, 70 thousand cases for Stage IV, and 31 thousand cases for Stage V in 2023.
- In Japan, there were approximately 73 thousand cases of psychosis associated with Parkinson’s disease in 2023.
- In the assessment done by DelveInsight, the estimated Diagnosed Prevalent Cases of MCI due to Parkinson’s Disease in the US were nearly 508 thousand in 2023.
- The leading Parkinson’s Disease Companies such as UCB Biopharma SRL, Novartis, Annovis Bio, Supernus Pharmaceuticals, Inc., Britannia Pharmaceutical, Pharma Two B, Mitsubishi Tanabe Pharma, NeuroDerm, AbbVie, Cerevel Therapeutics, LLC and others.
- Promising Parkinson’s Disease Therapies such as VTX3232, Rasagiline, Rotigotine, Levetiracetam, CVXL-0107, and others.
Stay ahead in the competitive landscape of the Parkinson’s Disease Market. Access DelveInsight’s in-depth market analysis and strategic insights today! Click here for more @ Parkinson’s Disease Treatment Market Size
Parkinson’s Disease Epidemiology Segmentation in the 7MM
- Total Diagnosed Prevalent Cases of Parkinson’s Disease
- Age-specific Diagnosed Prevalent Cases of Parkinson’s Disease
- Stage-specific Diagnosed Prevalent Cases of Parkinson’s Disease
- Gender-specific Diagnosed Prevalent Cases of Parkinson’s Disease
Download the report to understand which factors are driving Parkinson’s Disease epidemiology trends @ Parkinson’s Disease Prevalence
Parkinson’s Disease Marketed Drugs
- CREXONT (carbidopa and levodopa) ER capsules: Amneal Pharmaceuticals
CREXONT is a novel oral formulation of carbidopa/levodopa (CD/LD) that combines immediate-release (IR) granules and extended-release (ER) pellets. This prescription medication is used to treat Parkinson’s disease, Parkinson’s disease caused by brain infection or inflammation, and Parkinson’s disease-like symptoms resulting from carbon monoxide or manganese poisoning in adults. The ER beads in CREXONT consist of levodopa (LD), coated with a sustained-release polymer for slow drug release, a mucoadhesive polymer to ensure the granules remain at the absorption site longer, and an enteric coating to prevent premature disintegration in the stomach.
- PRODUODOPA (foslevodopa/foscarbidopa): AbbVie
PRODUODOPA is the first and only subcutaneous 24-hour infusion of levodopa-based therapy for treating severe motor fluctuations in advanced Parkinson’s disease patients whose symptoms are inadequately controlled by other therapies. The continuous delivery of PRODUODOPA provides levodopa 24 hours a day, potentially extending the period of well-controlled symptoms, often referred to as “On” time.
- XADAGO/EQUFINA (safinamide): Newron Pharmaceuticals/ Zambon/ Supernus Pharmaceuticals/ Eisai
XADAGO is an inhibitor of monoamine oxidase B (MAO-B) indicated as an adjunctive treatment to levodopa/carbidopa in patients with Parkinson’s disease experiencing “off” episodes. The precise mechanism by which XADAGO exerts its effect on Parkinson’s disease is unknown. However, it is believed that by inhibiting MAO-B activity and blocking the catabolism of dopamine, there is an increase in dopamine levels and subsequent dopaminergic activity in the brain. XADAGO tablets contain safinamide as the mesylate salt and are available as 50 mg and 100 mg film-coated tablets for oral administration.
Parkinson’s Disease Emerging Drugs
- SPN-830 (apomorphine infusion device): Supernus Pharmaceutical/Britannia Pharmaceuticals
SPN-830 (apomorphine infusion device) is an investigational apomorphine infusion device under review for the continuous treatment of motor fluctuations (OFF episodes) in Parkinson’s disease patients that are not adequately controlled with oral levodopa and one or more adjunct Parkinson’s disease medications. SPN-830 is an SC formulation of apomorphine delivered continuously through a portable electronic infusion pump under the skin for 14–18 h. The target infusion rate ranged between 3 mg and 8 mg per h.
- Tavapadon: AbbVie (Cerevel Therapeutics)
Cerevel Therapeutics is developing tavapadon to treat both early and late-stage Parkinson’s disease. Tavapadon was rationally designed as an orally bioavailable, once-daily partial agonist that selectively targets dopamine D1/D5 receptor subtypes to balance meaningful motor activity with a favourable safety profile.
- P2B001 (extended-release pramipexole and rasagiline): Pharma Two B
P2B001, being developed by Pharma Two B, is a novel, once-daily combination of extended-release pramipexole, a low-dose dopamine agonist, and rasagiline, a low-dose MAO-B inhibitor for the treatment of Parkinson’s disease. Pramipexole mimics the effects of dopamine while minimizing the breakdown of dopamine.
Discover the future of Parkinson’s Disease treatments with DelveInsight’s latest market report. Get expert insights and forecasts—download now! @ Parkinson’s Disease Market Drivers and Barriers
Parkinson’s Disease Market Outlook
Parkinson’s disease is a progressively worsening neurodegenerative disorder with an unclear cause and pathogenesis. Its global burden is increasing, driven by aging populations and environmental factors. Classic symptoms include motor issues like bradykinesia, rigidity, tremors, and postural instability, along with non-motor symptoms such as depression, cognitive impairment, and autonomic disturbances, which significantly affect quality of life. Diagnosing Parkinson’s is challenging due to symptom overlap with other movement disorders, with imaging like PET scans aiding in diagnosis.
Scope of the Parkinson’s Disease Market Report
- Coverage- 7MM
- Study Period- 2020-2034
- Parkinson’s Disease Companies- UCB Biopharma SRL, Novartis, Annovis Bio, Supernus Pharmaceuticals, Inc., Britannia Pharmaceutical, Pharma Two B, Mitsubishi Tanabe Pharma, NeuroDerm, AbbVie, Cerevel Therapeutics, LLC and others.
- Parkinson’s Disease Therapies- VTX3232, Rasagiline, Rotigotine, Levetiracetam, CVXL-0107, and others.
- Parkinson’s Disease Therapeutic Assessment: Parkinson’s Disease Current Marketed and Parkinson’s Disease Emerging Therapies
- Parkinson’s Disease Market Dynamics: Parkinson’s Disease market drivers and Parkinson’s Disease market barriers
- Parkinson’s Disease Unmet Needs, KOL’s views, Analyst’s views, Parkinson’s Disease Market Access and Reimbursement
Explore the dynamics of the Parkinson’s Disease Market with DelveInsight. From market size to emerging drugs—find it all in our latest report. Read now! @ Parkinson’s Disease Ongoing Clinical Trials Analysis
Table of Content
1. Key Insights
2. Report Introduction
3. Parkinson’s disease Market Overview at a Glance
4. Methodology of Parkinson’s disease Epidemiology and Market
5. Executive Summary of Parkinson’s Disease
6. Key Events
7. Disease Background and Overview
8. Epidemiology and Patient Population
9. Patient Journey
10. Marketed Therapies
11. Emerging Drug Profiles
12. Parkinson’s disease: Market Analysis
13. Key Opinion Leaders’ Views
14. SWOT Analysis
15. Unmet Needs
16. Market Access and Reimbursement
17. Appendix
18. DelveInsight Capabilities
19. Disclaimer
20. About DelveInsight
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Yash Bhardwaj
Email: Send Email
Phone: 09650213330
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/report-store/nontuberculous-mycobacterial-infections-market
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Parkinson’s Disease Treatment Market Size in the US was ~USD 1,883 million in 2023 and it is expected to grow by 2034 | DelveInsight