Menopause Companies are GlaxoSmithKline, Pfizer, Centro A.F. de Estudios Tecnologicos, S.A., IBSA Institut Biochimique SA, Massachusetts General Hospital, Watson Pharmaceuticals, Ausio Pharmaceuticals, LLC, Bayer, TherapeuticsMD, Organon and Co, Duramed Research, Noema Pharma AG, Unimed Pharmaceuticals, and others
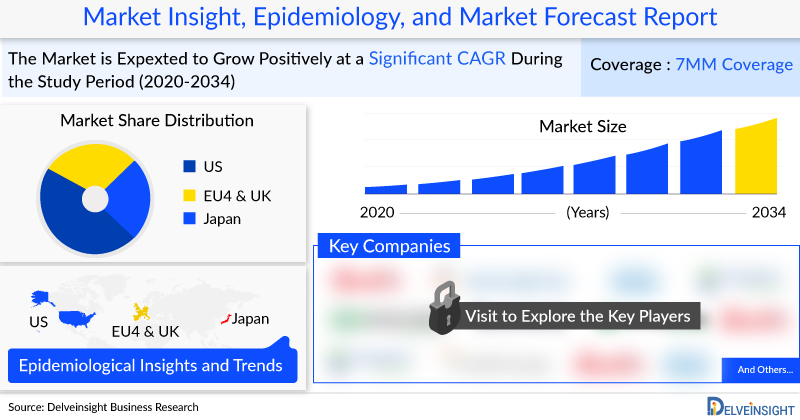
(Albany, USA) DelveInsight’s “Menopause Market Insights, Epidemiology, and Market Forecast-2034″ report offers an in-depth understanding of the Menopause, historical and forecasted epidemiology as well as the Menopause market trends in the United States, EU4 (Germany, Spain, Italy, France) the United Kingdom and Japan.
The latest healthcare forecast report provides an in-depth analysis of Menopause, offering comprehensive insights into the Menopause revenue trends, prevalence, and treatment landscape. The report delves into key Menopause statistics, highlighting the current and projected market size, while examining the efficacy and development of emerging Menopause therapies. Additionally, we cover the landscape of Menopause clinical trials, providing an overview of ongoing and upcoming studies that are poised to shape the future of Menopause treatment. This report is an essential resource for understanding the market dynamics and the evolving therapeutic options within the Menopause space.
To Know in detail about the Menopause market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Menopause Market Forecast
Some of the key facts of the Menopause Market Report:
-
The Menopause market size was valued ~USD 10,968 million in 2022 and is anticipated to grow with a significant CAGR during the study period (2020-2034)
-
In 2022, hormonal therapy accounted for approximately 63% of the total market share in the 7MM. However, its market share is expected to decline by 2034 as non-hormonal therapies become more prevalent.
-
Key Menopause Companies: GlaxoSmithKline, Pfizer, Centro A.F. de Estudios Tecnologicos, S.A., IBSA Institut Biochimique SA, Massachusetts General Hospital, Watson Pharmaceuticals, Ausio Pharmaceuticals, LLC, Bayer, TherapeuticsMD, Organon and Co, Duramed Research, Noema Pharma AG, Unimed Pharmaceuticals, and others
-
Key Menopause Therapies: GSK232802, DVS-233, Estradiol and Progesterone, Progesterone, Paroxetine, Premarin® (oral) vs. Alora® (transdermal), S-equol, LNG IUS, Estradiol, tibolone, DR-2041a, NOE-115, MELSMON, and others.
-
In May 2023, The US FDA recently approved VEOZAH (fezolinetant), a non-hormonal treatment that has demonstrated a significant reduction in the frequency and severity of moderate-to-severe VMS. VEOZAH has received approval in the United States, is under review in Europe, and is being introduced to the emerging market in Japan.
-
In 2022, the total number of menopause cases in the 7MM was 100,133,055. This number is expected to decline during the forecast period (2023-2034), primarily due to a decreasing female geriatric population in most of the countries within the 7MM.
-
In the 7MM, the stages of menopause among women were generally classified into perimenopausal (ages 40–65), postmenopausal (ages 40–65), and premature menopause (under 40 years).
-
In 2022, hot flashes were the most commonly observed symptom among women in menopause across the 7MM, making it the predominant condition during this stage of life.
-
In 2022, the total number of women in menopause across the EU4 (France, Germany, Italy, and Spain) and the UK was recorded at 41,300,409 cases, with projections indicating a decline in the coming years.
-
According to DelveInsight’s estimates, there were around 29,751,360 cases of hot flashes and 28,121,149 cases of night sweats in 2022, with these numbers expected to increase by 2034 during the study period.
-
The current selection of drugs for menopause includes a wide variety of medications. Notable emerging therapies in this area include DONESTA, BAY3427080, and VEOZAH, among others. Ongoing research and clinical trials have the potential to significantly impact the menopause treatment market.
Menopause Overview
Menopause is a natural biological process marking the end of a woman’s reproductive years. It occurs when the ovaries stop producing eggs, leading to a decline in estrogen and progesterone hormone levels. This transition typically happens between the ages of 45 and 55 but can occur earlier or later.
Menopause is confirmed after 12 consecutive months without a menstrual period. Common symptoms include hot flashes, night sweats, mood changes, sleep disturbances, and dryness.
Get a Free sample for the Menopause Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/menopause-market-forecast
Menopause Epidemiology
The epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Menopause Epidemiology Segmentation:
The Menopause market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
-
Total Number of Women in Menopause in the 7MM
-
Stage-specific Distribution of Women in Menopause in the 7MM
-
Age-specific Distribution of Women in Menopause in the 7MM
-
Number of Women in Menopause with Moderate-to-severe Vasomotor Symptoms in the 7MM
-
Number of Women in Menopause with Vasomotor Symptoms in the 7MM
Download the report to understand which factors are driving Menopause epidemiology trends @ Menopause Epidemiology Forecast
Menopause Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Menopause market or expected to get launched during the study period. The analysis covers Menopause market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Menopause Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Menopause Therapies and Key Companies
-
GSK232802: GlaxoSmithKline
-
DVS-233: Pfizer
-
Estradiol and Progesterone: Centro A.F. de Estudios Tecnologicos, S.A.
-
Progesterone: IBSA Institut Biochimique SA
-
Paroxetine: Massachusetts General Hospital
-
Premarin® (oral) vs. Alora® (transdermal): Watson Pharmaceuticals
-
S-equol: Ausio Pharmaceuticals, LLC
-
LNG IUS: Bayer
-
Estradiol: TherapeuticsMD
-
tibolone: Organon and Co
-
DR-2041a: Duramed Research
-
NOE-115: Noema Pharma AG
-
tibolone: Organon and Co
-
MELSMON: Unimed Pharmaceuticals
Discover more about therapies set to grab major Menopause market share @ Menopause Treatment Landscape
Menopause Market Strengths
Menopause Market Opportunities
Scope of the Menopause Market Report
-
Study Period: 2020–2034
-
Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
-
Key Menopause Companies: GlaxoSmithKline, Pfizer, Centro A.F. de Estudios Tecnologicos, S.A., IBSA Institut Biochimique SA, Massachusetts General Hospital, Watson Pharmaceuticals, Ausio Pharmaceuticals, LLC, Bayer, TherapeuticsMD, Organon and Co, Duramed Research, Noema Pharma AG, Unimed Pharmaceuticals, and others
-
Key Menopause Therapies: GlaxoSmithKline, Pfizer, Centro A.F. de Estudios Tecnologicos, S.A., IBSA Institut Biochimique SA, Massachusetts General Hospital, Watson Pharmaceuticals, Ausio Pharmaceuticals, LLC, Bayer, TherapeuticsMD, Organon and Co, Duramed Research, Noema Pharma AG, Unimed Pharmaceuticals, and others
-
Menopause Therapeutic Assessment: Menopause current marketed and Menopause emerging therapies
-
Menopause Market Dynamics: Menopause market drivers and Menopause market barriers
-
Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
-
Menopause Unmet Needs, KOL’s views, Analyst’s views, Menopause Market Access and Reimbursement
To know more about Menopause companies working in the treatment market, visit @ Menopause Clinical Trials and Therapeutic Assessment
Table of Contents
1. Menopause Market Report Introduction
2. Executive Summary for Menopause
3. SWOT analysis of Menopause
4. Menopause Patient Share (%) Overview at a Glance
5. Menopause Market Overview at a Glance
6. Menopause Disease Background and Overview
7. Menopause Epidemiology and Patient Population
8. Country-Specific Patient Population of Menopause
9. Menopause Current Treatment and Medical Practices
10. Menopause Unmet Needs
11. Menopause Emerging Therapies
12. Menopause Market Outlook
13. Country-Wise Menopause Market Analysis (2020–2034)
14. Menopause Market Access and Reimbursement of Therapies
15. Menopause Market Drivers
16. Menopause Market Barriers
17. Menopause Appendix
18. Menopause Report Methodology
19. DelveInsight Capabilities
20. Disclaimer
21. About DelveInsight
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Ankit Nigam
Email: Send Email
Phone: +14699457679
Address:304 S. Jones Blvd #2432
City: Albany
State: New York
Country: United States
Website: https://www.delveinsight.com/consulting
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Menopause Drugs Market 2034: Clinical Trials, EMA, PDMA, FDA Approvals, Medication, Epidemiology, Therapies, Companies by DelveInsight | Bayer, GSK, Pfizer, Centro A.F. de Estudios Tecnologicos, S.A.,