The Key Major Depressive Disorder Companies in the market include – Takeda Pharmaceuticals, Forest Laboratories, Otsuka Pharmaceuticals, Janssen Research & Development, Axsome Therapeutics, AbbVie, SAGE Therapeutics, Minerva Neurosciences, Luye Pharma, Relmada Therapeutics, BioLite Inc., VistaGen Therapeutics, Praxis Precision Medicines, Intra-Cellular Therapies, and others.
DelveInsight’s “Major Depressive Disorder Market Insights, Epidemiology, and Market Forecast-2034″ report offers an in-depth understanding of the Major Depressive Disorder, historical and forecasted epidemiology as well as the Major Depressive Disorder market trends in the United States, EU5 (Germany, Spain, Italy, France, and United Kingdom) and Japan.
To Know in detail about the Major Depressive Disorder market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Major Depressive Disorder Market Forecast
Key Highlights from Major Depressive Disorder market Report:
-
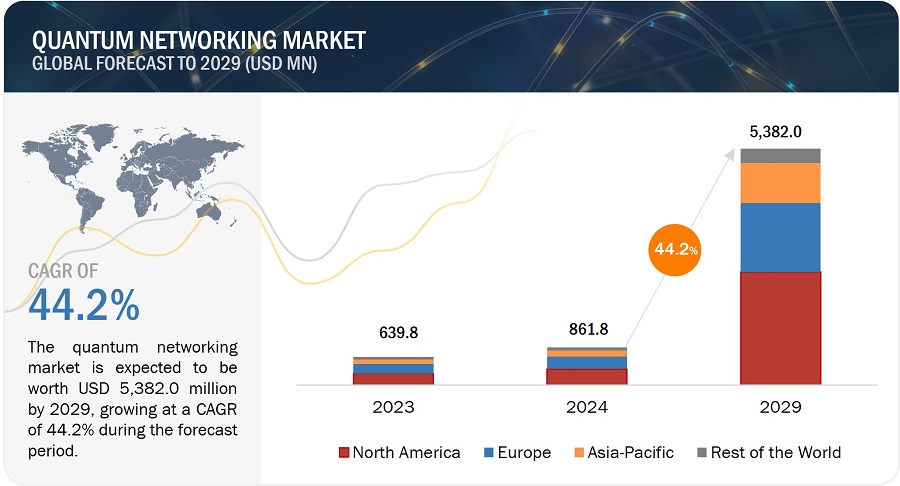
The Major Depressive Disorder market size was valued approximately USD 7,165 million in 2023 and is anticipated to grow with a significant CAGR during the study period (2020-2034)
-
In October 2024, Supernus Pharmaceuticals announced that its candidate SPN-820 showed a “rapid and significant” impact in a Phase IIa trial for adults with major depressive disorder (MDD). The study (NCT06235905) achieved its primary endpoint, demonstrating a clinically significant improvement of -6.1 on the Hamilton Depression Rating Scale-6 items (HAM-D6) at two hours, which increased to -9.6 by day 10. Additionally, suicidal ideation decreased by 80%, dropping from 12.5% at baseline to 2.6% by day ten.
-
In June 2024, MindBio Therapeutics, based in Vancouver, reported that its LSD-based drug, MB22001, has alleviated symptoms of depression in a Phase IIa trial. The durability data indicates that patients experienced a lasting response after self-administering a micro-dose of LSD. One month after stopping treatment, participants maintained a 65% reduction in depressive symptoms.
-
In April 2024, Alto Neuroscience began a Phase II clinical trial to evaluate the histamine H3 receptor inverse agonist, ALTO-203, for the treatment of major depressive disorder (MDD). This double-blind study will examine the pharmacodynamics, safety, tolerability, and pharmacokinetics of two doses of ALTO-203 monotherapy compared to a placebo, using both single and multiple doses.
-
In 2023, the United States held the largest share of the Major Depressive Disorder Treatment Market, representing about 78% of the total market size across the 7MM. This was significantly higher than the market sizes in other key regions, including the EU4 countries (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
-
In 2023, Germany held the largest Major Depressive Disorder market size among European countries, reaching approximately USD 531 million, whereas Italy had the smallest market size for Major Depressive Disorder at around USD 129 million.
-
Japan’s Major Depressive Disorder market size was estimated at approximately USD 142 million in 2023, representing 2% of the total market across the 7MM.
-
DelveInsight’s estimates indicate that the total diagnosed prevalent cases of Major Depressive Disorder in the 7MM were approximately 44 million in 2023.
-
In 2023, the United States had the highest number of diagnosed prevalent cases of Major Depressive Disorder, totaling around 22 million, with numbers anticipated to increase in the future.
-
In 2023, Germany had the highest number of diagnosed prevalent cases of Major Depressive Disorder among European countries, with approximately 6.8 million cases, followed by the UK with around 4 million cases. Conversely, Spain had the lowest diagnosed prevalence, with about 1.9 million cases.
-
In 2023, Japan had an estimated 3 million diagnosed prevalent cases of Major Depressive Disorder, representing about 7% of the total in the 7MM.
-
Key Major Depressive Disorder Companies: Takeda Pharmaceuticals, Forest Laboratories, Otsuka Pharmaceuticals, Janssen Research & Development, Axsome Therapeutics, AbbVie, SAGE Therapeutics, Minerva Neurosciences, Luye Pharma, Relmada Therapeutics, BioLite Inc., VistaGen Therapeutics, Praxis Precision Medicines, Intra-Cellular Therapies, and others
-
Key Major Depressive Disorder Therapies: VRAYLAR (Cariprazine), Zuranolone (SAGE-217/BIIB125), Seltorexant (JNJ42847922), SAGE-217, REL-1017, SP-624, SPL026, PDC-1421, and others
-
The Major Depressive Disorder epidemiology based on gender analyzed that females were more affected as compared to males in the United States
-
The Major Depressive Disorder market is expected to surge due to the disease’s increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Major Depressive Disorder pipeline products will significantly revolutionize the Major Depressive Disorder market dynamics.
Major Depressive Disorder Country based Treatment Overview:
The DelveInsight Market Research Report on Major Depressive Disorder provides a comprehensive insight into this condition, encompassing disease definition, symptoms, causes, pathophysiology, and diagnosis. It details both traditional and contemporary medical treatments and diagnostic options within the major depressive disorder market aimed at managing the condition.
Major Depressive Disorder (MDD), commonly known as depression, is a mental health condition characterized by persistent feelings of sadness, worthlessness, loss of interest or pleasure in activities, and changes in sleep, appetite, or energy levels. It significantly affects a person’s thoughts, feelings, behavior, and overall well-being, often interfering with daily functioning, work, and relationships.
Get a Free sample for the Major Depressive Disorder Market Report:
https://www.delveinsight.com/report-store/major-depressive-disorder-market
Major Depressive Disorder Epidemiology Insights:
The epidemiology section dedicated to major depressive disorder offers insights into the historical and present patient populations for this condition, along with projected trends across seven major countries. This section aids in understanding the reasons behind current and predicted trends by examining multiple studies and insights from key opinion leaders.
Major Depressive Disorder Epidemiology Segmentation:
The Major Depressive Disorder market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
-
Total Prevalence of Major Depressive Disorder
-
Prevalent Cases of Major Depressive Disorder by severity
-
Gender-specific Prevalence of Major Depressive Disorder
-
Diagnosed Cases of Episodic and Chronic Major Depressive Disorder
Download the report to understand which factors are driving Major Depressive Disorder epidemiology trends @ Major Depressive Disorder Epidemiology Forecast
Major Depressive Disorder Therapies and Key Companies
-
VRAYLAR (Cariprazine): AbbVie and Gedeon Richter Plc.
-
Zuranolone (SAGE-217/BIIB125): Sage Therapeutics/Biogen Inc., /Shionogi
-
Seltorexant (JNJ42847922): Janssen Pharmaceuticals/Minerva Neuroscience
-
SAGE-217: Sage Therapeutics
-
REL-1017: Relmada Therapeutics, Inc
-
SP-624: Sirtsei Pharmaceuticals, Inc.
-
SPL026: Small Pharma
-
PDC-1421: BioLite Inc
Emerging Major Depressive Disorder drugs Uptake:The pharmaceutical section within the major depressive disorder report comprises an in-depth analysis of marketed drugs for this condition as well as drugs in advanced stages (Phase-III, Phase-II/III, Phase-II, and Phase-I/II) within the major depressive disorder pipeline.
“The Phase II trial demonstrated a positive safety profile, good tolerability, and promising pharmacokinetics of REL-1017. Currently, it is undergoing advanced-stage development in Phase III trials for treating Major Depressive Disorder, both as an adjunctive therapy and as a standalone treatment.”
Discover more about therapies set to grab major Major Depressive Disorder market share @ Major Depressive Disorder Treatment Landscape
Major Depressive Disorder Market Forecast:
The market perspective presented in the report on major depressive disorder develops a comprehensive understanding of historical, present, and projected trends in the major depressive disorder market. It achieves this by assessing the influence of existing therapies, identifying unmet needs within Major Depressive Disorder, and evaluating the demand for advanced technology.
The Major Depressive Disorder market drivers include – there is an increasing awareness that Major Depressive Disorder can be reliably diagnosed and treated in primary care settings using antidepressant medications and/or brief structured psychological therapies.
Scope of the Major Depressive Disorder Market Report
-
Study Period: 2020–2034
-
Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
-
Key Major Depressive Disorder Companies: Takeda Pharmaceuticals, Forest Laboratories, Otsuka Pharmaceuticals, Janssen Research & Development, Axsome Therapeutics, AbbVie, SAGE Therapeutics, Luye Pharma, Relmada Therapeutics, BioLite Inc., VistaGen Therapeutics, Praxis Precision Medicines, Intra-Cellular Therapies, and others
-
Key Major Depressive Disorder Therapies: VRAYLAR (Cariprazine), Zuranolone (SAGE-217/BIIB125), Seltorexant (JNJ42847922), SAGE-217, REL-1017, SP-624, SPL026, PDC-1421, and others
-
Major Depressive Disorder Therapeutic Assessment: Major Depressive Disorder current marketed and Major Depressive Disorder emerging therapies
-
Major Depressive Disorder Market Dynamics: Major Depressive Disorder market drivers and Major Depressive Disorder market barriers
-
Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
-
Major Depressive Disorder Unmet Needs, KOL’s views, Analyst’s views, Major Depressive Disorder Market Access and Reimbursement
To know more about Major Depressive Disorder companies working in the treatment market, visit @ Major Depressive Disorder Clinical Trials and Therapeutic Assessment
Table of Contents
1. Major Depressive Disorder Market Report Introduction
2. Executive Summary for Major Depressive Disorder
3. SWOT analysis of Major Depressive Disorder
4. Major Depressive Disorder Patient Share (%) Overview at a Glance
5. Major Depressive Disorder Market Overview at a Glance
6. Major Depressive Disorder Disease Background and Overview
7. Major Depressive Disorder Epidemiology and Patient Population
8. Country-Specific Patient Population of Major Depressive Disorder
9. Major Depressive Disorder Current Treatment and Medical Practices
10. Major Depressive Disorder Unmet Needs
11. Major Depressive Disorder Emerging Therapies
12. Major Depressive Disorder Market Outlook
13. Country-Wise Major Depressive Disorder Market Analysis (2019–2032)
14. Major Depressive Disorder Market Access and Reimbursement of Therapies
15. Major Depressive Disorder Market Drivers
16. Major Depressive Disorder Market Barriers
17. Major Depressive Disorder Appendix
18. Major Depressive Disorder Report Methodology
19. DelveInsight Capabilities
20. Disclaimer
21. About DelveInsight
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Media Contact
Company Name: DelveInsight
Contact Person: Gaurav Bora
Email: Send Email
Phone: +14699457679
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: Nevada
Country: United States
Website: https://www.delveinsight.com/
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Major Depressive Disorder Market Expected to Experience Major Growth by 2034, According to DelveInsight | Takeda, Otsuka Pharma, AbbVie, SAGE Therapeutics, Forest Laboratories, Axsome Therapeutics