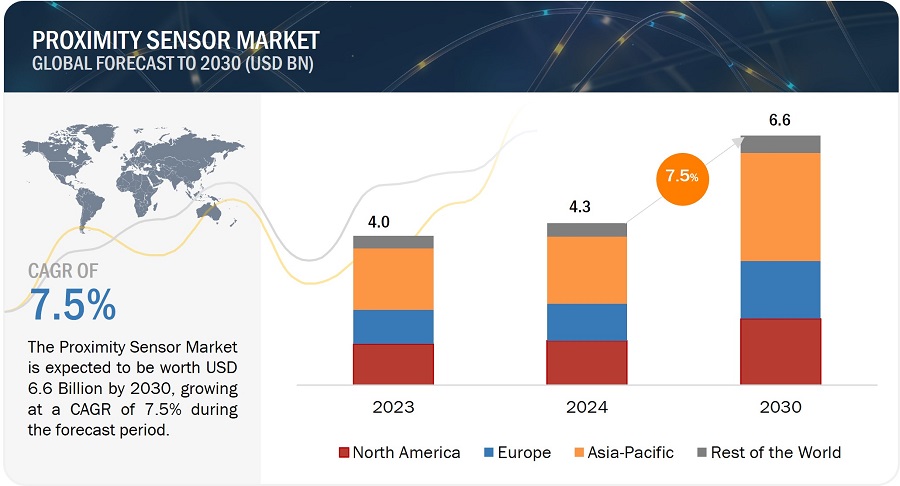
The report “Proximity Sensor Market by Technology (Inductive, Capacitive, Magnetic, Photoelectric/Optical, Ultrasonic), Product Type (Fixed distance, Adjustable distance), Range, Output, End-user Industry and Region – Global Forecast to 2030” The proximity sensor industry is projected to grow from USD 4.3 billion in 2024 and is estimated to reach USD 6.6 billion by 2030; it is expected to grow at a Compound Annual Growth Rate (CAGR) of 7.5% from 2024 to 2030.
The growth of the proximity sensor market is driven by the expansion of proximity sensors into new application areas such as medical devices, environmental monitoring, and smart city initiatives, Integration of proximity sensors with IoT and Al technology.
Download PDF Brochure @ https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=36281914
Browse 189 market data Tables and 73 Figures spread through 295 Pages and in-depth TOC on “Proximity Sensor Industry”
View detailed Table of Content here – https://www.marketsandmarkets.com/Market-Reports/proximity-sensor-market-36281914.html
By technology, Ultrasonic technology is projected to grow at a high CAGR of proximity sensor industry during the forecast period.
Ultrasonic proximity sensors emit a sound pulse that bounces back to the sensor from a target and generates an output signal. These sensors can detect metallic and non-metallic, transparent or opaque, liquid and solid objects offering effective enough acoustic reflectivity. Compared to photoelectric sensors, they are little affected by condensing moisture and hence apply in many environments. Rockwell Automation, Hans Turck GmbH & Co. The major companies operating in the market of ultrasonic proximity sensors include KG, TIMES France, and Migration Corp. The demand from the automotive sector has increased drastically due to the rising installation of ADAS in vehicles. Ultrasonic proximity sensors find high usage in improving vehicle safety and convenience, such as parking assistance, blind spot detection, and collision avoidance.
Adjustable distance segment in Product Type will account for highest CAGR during the forecast period.
Adjustable distance proximity sensors are ultra-modern devices designed to detect and measure the distance between the sensor and an object with adjustable precision. These sensors shall be using technologies like:
It gives accurate proximity sensing through ultrasonic, infrared, and laser-based systems across many industries; it is a sure deal in allowing automation, enhancing safety, and improving efficiency in their diversity of applications. Those companies located in Germany such as Pepperl+Fuchs; Japan, Omron Corporation; Germany, Sick AG; and the US-based Rockwell Automation have developed adjustable distance proximity sensors that detect metallic and non-metallic objects within an adjustable sensing range. This feature enables its application where sensing range might vary, like full or empty tank detection. Proximity sensors with an adjustable distance option are used more in comparison to fixed distance proximity sensors, which detect only at a specified distance.
In applications that require a high degree of precision and sensitivity, proximity sensors with a range below 10 mm become essential. Detection of the presence or absence of objects at very short distances makes the sensor quite suitable where minute accuracy is required. Consumer electronics, medical devices, and the automotive industry are the industries that need these sensors immensely for the simple reason that they can function in infinitesimal spaces, even tracing minute movements. Since the invention of IoT devices, need-based requirements for compact and efficient sensors in consumer electronics have increased manifold. These short-range proximity sensors find places in wireless earbuds, smart watches, and VR headsets for attachment/detachment detection.
Analog Proximity Sensor in Output segment in proximity sensor industry will account for largest market share in the forecast timeline.
Compared with digital proximity sensors, analog ones also have advantages in high-precision and continuous-monitoring applications. Where digital sensors will give a binary output, analog sensors give variable output, which changes linearly with distance detection. This becomes very critical in tasks that require fine-tuned control and accurate distance measurements, like robotic arm positioning, conveyor belt speed regulation, or quality control during manufacturing processes. It is in this way that technologies of Industry 4.0, focusing on automation, connectivity, and data exchange, increase the demand for such sensors at an incredible rate.
Automotive in end-user industry segment in proximity sensor industry will account for the highest CAGR by 2030.
With rising demands for advanced safety and, more recently, autonomous driving features, vehicle manufacturers across the globe are seeking proximity sensors. For instance, in June 2023, Leuze introduced the HTU200 and DMU200 series of highly compact ultrasonic products with applications oriented to the packaging and automobile industries. Such sensors detect objects precisely and independently of their surface quality: glossy and transparent surfaces and at distances between 0.1 and 6 meters. Large companies making automotive, like Tesla, BMW, and Audi, increase the diffusion of the design of vehicles, setting examples for others to follow. Proximity sensors will probably play a more significant role in the industry’s future when technology develops, and vehicle sophistication increases.
Asia Pacific will account for the highest CAGR during the forecast period.
As industries aim to achieve efficient operations, thereby reducing downtime and enhancing the quality of their products, proximity sensors, which make all these advancements possible in automation and safety protocols, will likely extend their demand manifold in the near future. Countries like China and South Korea account for the lion’s share in the electronics manufacturing world; hence, demands for proximity sensors in this segment are strong. Proximity sensors are applied in electronic assembly lines for component detection, checking correct placement, and enhancing production efficiency. The growth of consumer electronics market in Asia Pacific, driven by demand from an increasing disposable income and technological development, further fuels the demand for these sensors. For instance, China is the largest manufacturer of electronics globally, thus offering significant regional demand for proximity sensors.
Key Players
Key companies operating in the proximity sensor industry are OMRON Corporation (Japan), KEYENCE CORPORATION (Japan), Panasonic Corporation (Japan), SICK AG (Germany), STMicroelectronics (Switzerland), Rockwell Automation (US), Delta Electronics, Inc. (Taiwan), Honeywell International Inc. (US), Broadcom (US), Schneider Electric (France), ZF Friedrichshafen AG (Germany), Balluff Automation India Pvt. Ltd. (India), Pepperl+Fuchs SE (Germany), Baumer (Switzerland), Banner Engineering Corp. (US), among others.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103
City: Delray Beach
State: FL 33445
Country: United States
Website: https://www.marketsandmarkets.com/Market-Reports/proximity-sensor-market-36281914.html
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Proximity Sensor Market worth $6.6 Billion by 2030, at a CAGR of 7.5%