Key Acute Respiratory Distress Syndrome companies are MediciNova, Edesa Biotech, Light Chain Biosciences, Boehringer Ingelheim, Genentech, Windtree Therapeutics, Biomarck Pharmaceuticals, Athersys, Healios, Direct Biologics, Biohaven Pharmaceutical, Arch Biopartners, APEPTICO Forschung und Entwicklung GmbH, Staidson (Beijing) Biopharmaceuticals, Veru, Mesoblast Limited, Avalo Therapeutics, Pluristem Therapeutics, ILTOO Pharma, and others.
Expected launch of emerging therapies, such as Traumakine (Faron Pharmaceuticals) BIO-11006 (BioMarck Pharmaceuticals), MultiStem (Athersys), Solnatide (Apeptico Forschung und Entwicklung GmbH), PB1046 (PhaseBio Pharmaceuticals), and others are expected to create a positive impact on the ARDS treatment scenario and ARDS market outlook in the upcoming years.
DelveInsight’s “Acute Respiratory Distress Syndrome Market Insights, Epidemiology, and Market Forecast 2034” report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the Acute Respiratory Distress Syndrome market size, share, trends, and growth opportunities in the seven major markets (7MM) (i.e., the United States, EU4 (Germany, Spain, Italy, France), the United Kingdom and Japan).
The Acute Respiratory Distress Syndrome market report covers emerging drugs, current treatment practices, market share of individual therapies, and current & forecasted market size from 2020 to 2034. It also evaluates the current treatment practice/algorithm, key drivers & barriers impacting the market growth, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the market.
Key highlights from the Acute Respiratory Distress Syndrome Market Report:
-
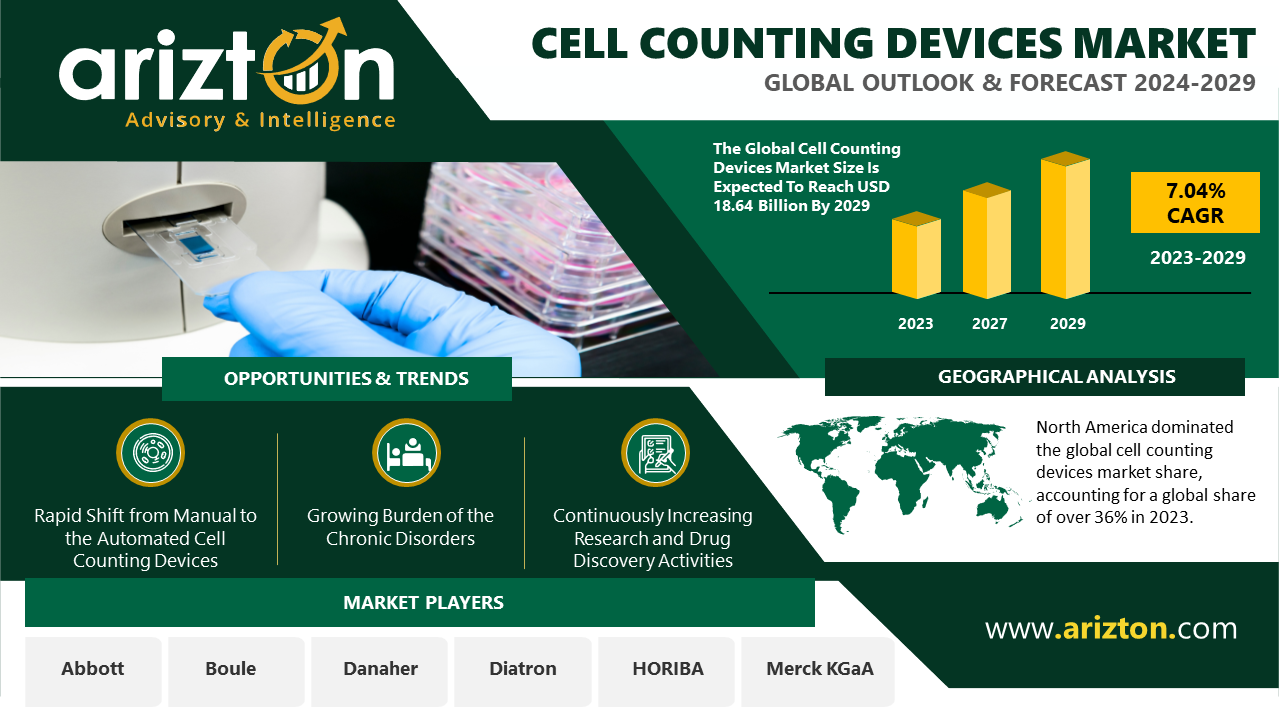
The market for acute respiratory distress syndrome (ARDS) in the 7MM was valued at approximately USD 941 million in 2023 and is projected to grow by 2034. Among the 7MM, the U.S. had the largest market share in 2023, accounting for around USD 666.9 million. This figure is expected to rise further by 2034 due to increasing rates of chronic health conditions and the emergence of new infectious diseases, including novel viral strains.
-
Several factors contribute to the rising incidence of ARDS. Environmental pollutants, such as air pollution and toxins, exacerbate respiratory issues. Additionally, chronic conditions like diabetes, heart disease, and obesity, especially when poorly managed, elevate the risk of developing ARDS.
-
The ARDS market is anticipated to experience steady growth from 2024 to 2034. This growth is driven by an aging population that is more susceptible to long-term illnesses, weakened immune systems, and an increased prevalence of viral and bacterial infections, including pneumonia, influenza, sepsis, and emerging coronavirus strains.
-
Current treatment strategies for ARDS include supportive care, mechanical ventilation, oxygen therapy, sedation, corticosteroids, ACE inhibitors, ARBs, neuromuscular blockers, inhaled vasodilators, and, in some cases, extracorporeal membrane oxygenation (ECMO). Supportive measures such as prone positioning, fluid management, and conservative fluid strategies are also used to optimize patient outcomes.
Read detailed insights on ARDS market outlook 2034 @ https://www.delveinsight.com/report-store/acute-respiratory-distress-syndrome-ards-market
Acute Respiratory Distress Syndrome Overview
Acute respiratory distress syndrome (ARDS) is a rapidly progressive condition that primarily affects critically ill patients. It is marked by the accumulation of fluid in the lungs, which severely impairs breathing and results in low blood oxygen levels. ARDS often arises as a complication of another serious illness or major injury, leading to fluid buildup in the lung’s tiny air sacs and a breakdown of surfactant.
The causes of ARDS are categorized into two main types: direct and indirect lung injuries. Direct lung injuries include conditions such as pneumonia, aspiration, and trauma. Indirect lung injuries involve systemic issues such as pancreatitis, severe infections (sepsis), blood transfusion reactions, burns, and adverse drug reactions.
The initial symptoms of ARDS typically include shortness of breath, cough, and fever. Other signs may involve low blood oxygen levels, rapid breathing, and abnormal lung sounds such as clicking, bubbling, or rattling.
ARDS is generally described in three stages:
1. Exudative Stage: Occurs within the first 2 to 4 days after lung injury, characterized by the accumulation of protein-rich fluid, excessive fluid, and inflammatory cells in the alveoli.
2. Fibroproliferative (Proliferative) Stage: This stage involves the proliferation of connective tissue and structural elements in response to lung injury. Patients are at higher risk for complications like pneumonia, sepsis, and lung rupture during this phase.
3. Resolution and Recovery Stage: During this phase, the lung begins to reorganize and recover, with potential improvements in lung function.
Some experts also identify a fourth phase, where patients may face long-term health issues or psychological effects similar to post-traumatic stress disorder (PTSD), including anxiety, depression, and flashbacks related to their critical illness.
Acute respiratory distress syndrome diagnosis
Early detection of acute respiratory distress syndrome (ARDS) is crucial for promptly initiating lung-protective ventilation strategies. Despite efforts to refine disease definitions, ARDS is often under-recognized by clinicians, with diagnoses frequently delayed. Reports suggest that up to 40% of ARDS cases are under-diagnosed, with diagnostic rates improving as the disease severity increases. ARDS diagnosis relies on several clinical criteria, none of which are particularly precise. For instance, the Berlin definition has a specificity of only 63% for detecting diffuse alveolar damage (DAD) at autopsy. Currently, no specific biomarker exists for ARDS diagnosis, making this a significant focus of ongoing research.
Chest X-rays (CXR) have relatively low sensitivity and specificity (around 70%) compared to CT scans for diagnosing ARDS. CXR is more effective in identifying diffuse or patchy infiltrates than focal ones. The use of CT scans and other imaging techniques has become increasingly important in diagnosing and managing ARDS. Additionally, lung ultrasound is gaining recognition as a useful bedside tool for facilitating ARDS diagnosis.
Acute Respiratory Distress Syndrome Treatment Market
The treatment of acute respiratory distress syndrome (ARDS) primarily involves supportive care. This includes mechanical ventilation, prevention of stress ulcers and venous thromboembolism, and nutritional support. Effective supportive care for ARDS patients, as with other ICU patients, encompasses early enteral feeding, good glycemic control, prevention of deep venous thrombosis, and prophylaxis against stress ulcers. It is also crucial to identify and treat any underlying infections with antibiotics tailored to culture sensitivities; if cultures are unavailable, antibiotics should target common pathogens relevant to the infection site.
Additional treatments for ARDS patients typically involve supplemental oxygen, prone positioning, the use of neuromuscular blockers (paralytics), fluid management, and positive end-expiratory pressure (PEEP) to help expel fluid from the air sacs. These supportive measures are combined with ongoing treatment of the primary illness or injury. Given that ARDS patients are more susceptible to lung infections, such as bacterial pneumonia, antibiotics are administered to manage these infections. Supportive care may also include intravenous fluids or nutrition as needed.
Acute Respiratory Distress Syndrome Epidemiology Assessment
The epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted epidemiology trends by exploring numerous studies and research. The epidemiology section also provides a detailed analysis of diagnosed and prevalent patient pool, future trends, and views of key opinion leaders.
Acute Respiratory Distress Syndrome Epidemiology Insights:
-
In 2023, DelveInsight estimated approximately 862,000 new cases of acute respiratory distress syndrome (ARDS) across the 7MM. This number is expected to rise over the forecast period from 2024 to 2034, largely due to environmental factors such as air pollution and toxin exposure, which exacerbate respiratory diseases.
-
In the U.S., there were about 545,000 new ARDS cases in 2023. This figure is anticipated to grow by 2034 due to the increasing prevalence of chronic conditions like diabetes, heart disease, and obesity.
-
Among the EU4 and the UK, Germany had the highest incidence of ARDS with approximately 157,000 cases in 2023, followed by Italy with around 45,000 cases. Spain had the lowest incidence, with about 21,000 cases.
-
In 2023, the EU4 and the UK reported approximately 44,000 cases of mild ARDS, 132,000 cases of moderate ARDS, and 122,000 cases of severe ARDS.
-
In Japan, there were about 19,000 new ARDS cases in 2023. The primary risk factors included pneumonia (34%), sepsis (29%), trauma (7%), aspiration (10%), pancreatitis (2%), other causes (16%), and unknown factors (2%).
Acute Respiratory Distress Syndrome Emerging Drugs
-
EB05 (paridiprubart): Edesa Biotech/Light Chain Biosciences
-
Alteplase (Actilyse): Boehringer Ingelheim/ Genentech
-
BIO-11006: BioMarck Pharmaceuticals
-
ExoFlo (DB-001): Direct Biologics
Learn How the Acute Respiratory Distress Syndrome Market Will Evolve and Grow by 2034 @ https://www.delveinsight.com/sample-request/acute-respiratory-distress-syndrome-ards-market
Acute Respiratory Distress Syndrome Market
Despite decades of research, treatment options for acute respiratory distress syndrome (ARDS) remain limited. The primary approach to managing ARDS continues to be supportive care with mechanical ventilation, as there are relatively few available treatments.
In addition to mechanical ventilation, ARDS management often includes supplemental oxygen, prone positioning, neuromuscular blockers (paralytics), fluid management, and the use of positive end-expiratory pressure (PEEP) to help remove fluid from the air sacs. These treatments are combined with ongoing management of the underlying illness or injury. ARDS patients are also at increased risk for bacterial pneumonia, so antibiotics are administered as needed. Supportive care may include intravenous fluids and nutrition.
Key pathophysiological issues in ARDS include alveolar flooding and pulmonary edema. Experimental studies have shown that β2 agonists can enhance sodium transport by activating β2 receptors on alveolar cells, potentially accelerating the resolution of pulmonary edema.
Alveolar epithelial injury is a significant contributor to ARDS, and accelerating the repair of these epithelial cells may help resolve pulmonary edema and lung injury. Keratinocyte growth factor (KGF) plays a crucial role in alveolar epithelial repair, and both experimental and clinical studies suggest that KGF could be beneficial for ARDS patients.
The U.S. FDA has issued an emergency use authorization (EUA) for Actemra/RoActemra (tocilizumab) for the treatment of hospitalized adults and pediatric patients (2 years and older) who are receiving systemic corticosteroids and need supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO). Additionally, the FDA has approved Pluristem Therapeutics’ Expanded Access Program (EAP) for using its PLX-PAD cells to treat ARDS caused by COVID-19, outside of the ongoing Phase II study. Current therapies have mainly addressed ARDS-related inflammation, with no specific treatments available for impaired lung function.
To address the significant unmet needs in ARDS treatment, companies worldwide are focusing on developing new therapies. Emerging treatments are anticipated to have a substantial impact on the ARDS market. These include EB05 (Edesa Biotech), BIO-11006 (BioMarck Pharmaceuticals), MultiStem (Athersys), Alteplase (Boehringer Ingelheim/Genentech), Lucinactant (Windtree Therapeutics), and DB-001 (Direct Biologics), which are expected to launch between 2024 and 2034.
According to DelveInsight, the ARDS market dynamics are expected to evolve in the coming years with the introduction of these new therapies.
Leading Players in the Acute Respiratory Distress Syndrome Therapeutics Market Include:
MediciNova, Edesa Biotech, Light Chain Biosciences, Boehringer Ingelheim, Genentech, Windtree Therapeutics, Biomarck Pharmaceuticals, Athersys, Healios, Direct Biologics, Biohaven Pharmaceutical, Arch Biopartners, APEPTICO Forschung und Entwicklung GmbH, Staidson (Beijing) Biopharmaceuticals, Veru, Mesoblast Limited, Avalo Therapeutics, Pluristem Therapeutics, ILTOO Pharma, and others.
Table of Contents
1. Key Insights
2. Executive Summary
3. Acute Respiratory Distress Syndrome Competitive Intelligence Analysis
4. Acute Respiratory Distress Syndrome Market Overview at a Glance
5. Acute Respiratory Distress Syndrome Disease Background and Overview
6. Acute Respiratory Distress Syndrome Patient Journey
7. Acute Respiratory Distress Syndrome Epidemiology and Patient Population (In the US, EU5, and Japan)
8. Acute Respiratory Distress Syndrome Treatment Algorithm, Current Treatment, and Medical Practices
9. Acute Respiratory Distress Syndrome Unmet Needs
10. Key Endpoints of Acute Respiratory Distress Syndrome Treatment
11. Acute Respiratory Distress Syndrome Marketed Products
12. Acute Respiratory Distress Syndrome Emerging Drugs and Latest Therapeutic Advances
13. Acute Respiratory Distress Syndrome Seven Major Market Analysis
14. Attribute Analysis
15. Acute Respiratory Distress Syndrome Market Outlook (In US, EU5, and Japan)
16. Acute Respiratory Distress Syndrome Access and Reimbursement Overview
17. KOL Views on the Acute Respiratory Distress Syndrome Market
18. Acute Respiratory Distress Syndrome Market Drivers
19. Acute Respiratory Distress Syndrome Market Barriers
20. Appendix
21. DelveInsight Capabilities
22. Disclaimer
Trending Reports by DelveInsight:
Adalimumab Biosimilar Market | Arbovirus Infection Market | Artificial Pancreas Device System Market | Dental Equipment Market | Gluten Sensitivity Market | Hypothyroidism Market | Inflammatory Bowel Disease Market | Mayus Kinase Jak Inhibitors Market | Mild Dry Eye Market | Mucopolysaccharidosis Market | Oncolytic Virus Cancer Therapy Market | Pyoderma Gangrenosum Market | Transdermal Drug Delivery Devices Market
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Kritika Rehani
Email: Send Email
Phone: +14699457679
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: Nevada
Country: United States
Website: https://www.delveinsight.com/
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Acute Respiratory Distress Syndrome Market size in 7MM was ~USD 941 million in 2023, asserts DelveInsight | MediciNova, Edesa Biotech, Light Chain Biosciences, Boehringer Ingelheim, Genentech