Food Fortifying Agents Market by Type (Minerals, Vitamins, Carbohydrates, Prebiotics, Probiotics), Application (Cereal & Cereal-based Products, Bulk Food Items), Process (Drum Drying, Dusting) and Region – Global Forecast to 2027
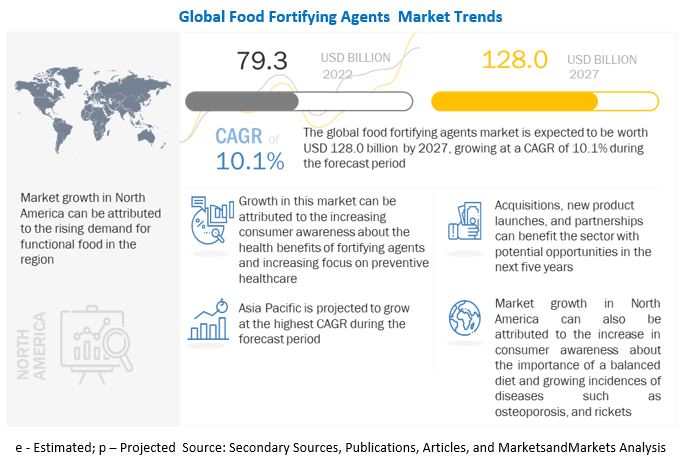
The global food fortifying agents market is estimated to reach $128.0 billion by 2027, growing at a 10.1% compound annual growth rate (CAGR). The global market size was valued $79.3 billion in 2022. With such remarkable figures on the horizon, the global market is undoubtedly one to watch out for.The food fortifying agents industry is poised for significant expansion in response to the growing emphasis on health and wellness among consumers. With a surge in demand for products that offer added nutritional benefits, there is a unique opportunity for innovation and growth in the market. As the aging population grows and health concerns become more prevalent, the adoption of food fortifying agents for a variety of applications is expected to increase on a large scale. This trend is set to drive the market forward, creating new avenues for manufacturers and suppliers to cater to the evolving needs of health-conscious consumers.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=72061350
The Evolution of the Food Fortifying Agents Market
Over the past few decades, the food fortifying agents market has experienced remarkable evolution. Initially, the process was straightforward, such as adding iodine to salt to prevent goiter. However, it has since developed into a sophisticated industry that produces a diverse array of additives to enhance foods with essential nutrients. This market growth is driven by several factors. A primary factor is the increasing awareness of the importance of a balanced diet and the critical role essential nutrients play in maintaining optimal health. Additionally, with the rising prevalence of chronic diseases like obesity and diabetes, consumers are increasingly seeking foods that are not only delicious but also beneficial for their health.
Increasing demand for fortifying agents in dietary supplements
The primary factor driving the demand for food fortifying agents is the increasing popularity of dietary supplements. As lifestyles evolve, consumer awareness about the benefits of dietary supplements has grown. There has been a significant rise in the consumption of vitamins, minerals, immunity-boosting foods, organic products, and various dietary aids. Higher disposable incomes and greater health consciousness among consumers have spurred the shift towards nutraceuticals, including dietary supplements. Consequently, more people are opting for multivitamins and minerals in capsule and tablet form due to lifestyle changes and a heightened focus on fitness and health.
The rising consumption of omega-3 fatty acids to reduce the risk if chronic heart diseases
Lipids are a diverse group of naturally occurring molecules that include waxes, fats, sterols, fat-soluble vitamins (such as vitamins A, D, E, and K), monoglycerides, triglycerides, diglycerides, and phospholipids. Among these, omega-3 and omega-6 fatty acids are essential lipids that are commonly used.
Omega-3 fatty acids, a type of polyunsaturated fatty acid (PUFA), are essential for health. They can be found in fish such as tuna, salmon, and halibut, as well as in algae, krill, certain plants, and nut oils. Consuming omega-3 fatty acids has been shown to reduce the risk of various health issues, including heart disease, prostate and breast cancers, high blood pressure, osteoporosis, bipolar disorder, depression, skin disorders, asthma, and arthritis. The American Heart Association recommends eating fish, especially fatty varieties like mackerel, lake trout, herring, sardines, albacore tuna, and salmon, at least twice a week to promote better health.
Make an Inquiry: https://www.marketsandmarkets.com/Enquiry_Before_BuyingNew.asp?id=72061350
Asia Pacific region is witnessing the highest growth rate in the market.
The food fortifying agents market in the Asia Pacific region is experiencing significant growth, driven by the busy lifestyles and increasing disposable incomes of consumers. The demand for these agents is expected to rise rapidly due to growing consumer interest. Among these agents, proteins and amino acids are expanding particularly quickly. Key markets in this region include China, India, Japan, and Australia & New Zealand, which boast extensive agricultural areas and produce high-fiber plants and oilseeds—primary raw materials for fiber and specialty carbohydrate ingredients.
The processed food industry in the Asia Pacific is also expanding, fueled by changing consumer lifestyles. The food and beverage sector is undergoing a substantial transformation, prompted by rapid urbanization, diet diversification, and the liberalization of foreign direct investment in the food industry. Increasing incomes and purchasing power, along with a rising demand for nutritional and healthy products, present promising opportunities for growth and diversification in the region’s food sector, further boosting the demand for food fortifying agents.
Top Companies in the Food Fortifying Agents Market
The key players in this market include Cargill (US), DSM (Netherlands), CHR. Hansen Holdings A/S (Denmark), DuPont (US), BASF SE (Germany), Arla Foods Amba (Denmark), Tate & Lyle (UK), Ingredion (US), Archer Daniels Midland Company (US), and Nestle SA (Switzerland).
Cargill is involved in the manufacturing and marketing of food ingredients, agricultural products, risk management, financial, and industrial products & services around the globe. Cargill operates in 70 countries through its subsidiaries and affiliates. The company manufactures and markets food fortifying agents such as oils & fats, grains & oil seeds. The company distributes its wide range of indigenous and imported vegetable oils, fats and flour products to household customers as well as food industries in over 125 countries at a global level. Its geographical network spans North America, Latin America, Europe, Asia Pacific, and Middle East & Africa. Its subsidiaries include The Mosaic Company (US), NatureWorks LLC (US), and The Wilbur Chocolate Company (US).
DSM is the one of the leading companies providing specialized solutions for health, nutrition & Bioscience. DSM offers a wide range of products for animal nutrition & health, human nutrition, care & health, food & beverage and materials. The company supplies food & beverage, dietary supplements, early-life nutrition and medical nutrition, whereas in animal nutrition & health is the leading supplier of feed additives such as vitamins, carotenoids, eubiotics, and feed enzymes. The company provides scientific & innovative solutions to 23,000 people throughout the world. The company has geographical presence across North America, Europe, Asia Pacific, Latin America, Netherlands, Switzerland and Rest of the World. Its subsidiaries include DSM Nutritional products AG, GLYCOM A/S, Fortitech, Inc., and Vitatene S.A.
Get 10% Free Customization on this Report: https://www.marketsandmarkets.com/requestCustomizationNew.asp?id=72061350
About MarketsandMarkets™
MarketsandMarketsTM has been recognized as one of America’s best management consulting firms by Forbes, as per their recent report.
MarketsandMarkets™ is a blue ocean alternative in growth consulting and program management, leveraging a man-machine offering to drive supernormal growth for progressive organizations in the B2B space. We have the widest lens on emerging technologies, making us proficient in co-creating supernormal growth for clients.
Earlier this year, we made a formal transformation into one of America’s best management consulting firms as per a survey conducted by Forbes.
The B2B economy is witnessing the emergence of $25 trillion of new revenue streams that are substituting existing revenue streams in this decade alone. We work with clients on growth programs, helping them monetize this $25 trillion opportunity through our service lines – TAM Expansion, Go-to-Market (GTM) Strategy to Execution, Market Share Gain, Account Enablement, and Thought Leadership Marketing.
Built on the ‘GIVE Growth’ principle, we work with several Forbes Global 2000 B2B companies – helping them stay relevant in a disruptive ecosystem. Our insights and strategies are molded by our industry experts, cutting-edge AI-powered Market Intelligence Cloud, and years of research. The KnowledgeStore™ (our Market Intelligence Cloud) integrates our research, facilitates an analysis of interconnections through a set of applications, helping clients look at the entire ecosystem and understand the revenue shifts happening in their industry.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:630 Dundee Road Suite 430
City: Northbrook
State: IL 60062
Country: United States
Website: https://www.marketsandmarkets.com/Market-Reports/food-fortifying-agents-market-72061350.html
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Food Fortifying Agents Market Trends, Growth Opportunities, Industry Overview, Top Companies, and Forecast – 2027