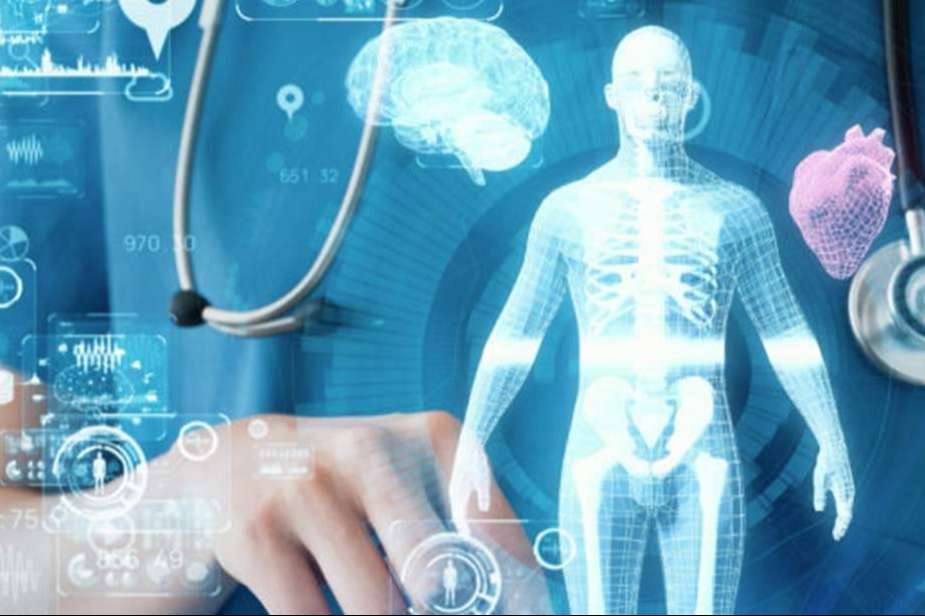
Innovation is the lifeblood of progress. In healthcare, it’s the key to improving patient outcomes and reducing costs
Enter iTera Bio Lite. This advanced technology is transforming the healthcare industry
But what exactly is iTera Bio Lite? It’s a tool that integrates seamlessly with existing healthcare systems. It aids in patient diagnosis and treatment, enhancing the quality of care.
The iTera-Bio Lite price is competitive, offering cost-effective solutions for healthcare facilities. It’s not just about the cost, though. The benefits of iTera-Bio Lite extend beyond financial savings.
In this article, people will delve into the world of iTera Bio Lite. People will explore how this healthcare innovation is shaping the future of medicine. Stay tuned to learn more about this game-changing technology.
Understanding healthcare innovation
Healthcare innovation refers to the application of new or improved solutions in the medical field. These solutions can be products, services, or processes that enhance healthcare delivery.
Innovation in healthcare is crucial. It helps to improve patient outcomes, reduce healthcare costs, and increase efficiency. It’s the driving force behind advancements in patient care and the overall healthcare system.
Introducing iTera-Bio Lite: A leap in healthcare technology
One such innovation making waves in the healthcare industry. This advanced technology is designed to transform patient care and healthcare delivery.
iTera Bio Lite integrates seamlessly with existing healthcare systems. It’s designed to enhance patient diagnosis and treatment, making it a valuable tool for healthcare professionals. The technology’s potential to improve healthcare is immense, and it’s already showing promising results in various medical settings.
The functionality of iTera Bio Lite
The iTera Bio Lite is more than just a piece of hardware. It’s a comprehensive solution that combines advanced technology with user-friendly interfaces. This makes it a practical tool for healthcare professionals across various specializations.
The technology operates with Terahertz waves to deliver an energizing, invigorating and soothing experience that promotes a comforting effect. It uses sinusoidal energy to energize and rejuvenate. It employs the graphene beam to facilitate relaxation and relieve tension. PEMF therapy harnesses technology to stimulate and exercise cells, addressing cellular dysfunction and supporting overall wellness.
TeraHertz
Sinusoidal Energy
Graphene far infrared ray
The adaptability of iTera-Bio Lite to different medical specializations makes it a versatile tool in the healthcare industry. Whether it’s for general practice or specialized care.
iTera Bio Lite Price Shop
Cost-Effectiveness: Analyzing iTera-Bio Lite price and healthcare savings
Investing in iTera-Bio Lite is a strategic decision for healthcare facilities. While the iTera-Bio Lite price may seem substantial initially, the long-term savings are considerable. The technology’s efficiency and accuracy can reduce unnecessary tests and procedures, leading to significant cost savings.
Moreover, iTera Bio Lite’s potential to improve patient outcomes can also contribute to financial savings. Better patient outcomes often mean shorter hospital stays and fewer readmissions, which can greatly reduce healthcare costs.
iTera-Bio Lite Benefits: Improving patient outcomes and care 1uality
The benefits of iTera Bio extend beyond cost savings. This innovative technology can significantly improve patient outcomes and the overall quality of care. iTera-Bio Lite helps healthcare technology professionals make informed decisions regarding patient care by providing accurate and timely data.
Some of the key benefits of iTera-Bio Lite include:
Enhanced diagnostic accuracy
Improved patient monitoring
Personalized treatment plans
Reduced risk of medical errors
In essence, iTera-Bio Lite is not just a technological innovation. It’s a tool that can transform patient care and contribute to the overall improvement of healthcare quality.
Conclusion: The Role of iTera-Bio Lite in shaping modern health tech
In conclusion, is more than just a healthcare innovation. It is a tool that has the potential to reshape modern healthcare. By improving diagnosis, treatment, and patient outcomes, iTera-Bio Lite is making a significant impact on healthcare delivery.
Moreover, the cost-effectiveness of iTera-Bio Lite makes it an attractive option for healthcare facilities. The iTera-Bio Lite price is competitive, and the potential savings in healthcare costs are substantial. This makes iTera-Bio Lite a smart investment for healthcare providers.
Looking ahead, iTera Bio Lite is set to play an even bigger role in healthcare. With continuous updates and developments, iTera Bio Lite is poised to stay at the forefront of healthcare innovation. This ensures that iTera Bio Lite will continue to contribute to the improvement of healthcare for years to come.
Media Contact
Company Name: ITera-Bio Lite
Contact Person: Press Office
Email: Send Email
Country: United States
Website: https://iteracare.life/en/