“Delveinsight Business Research LLP”
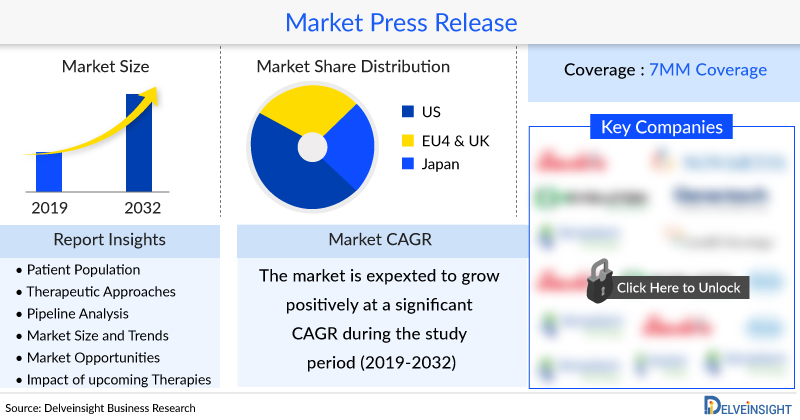
The Head and Neck Cancer Market in the 7MM was observed to be USD 2,801 million in 2021, which is expected to grow by 2032. As per DelveInsight, the Head and Neck Cancer Market is anticipated to evolve immensely in the coming years owing to the increase in the patient pool and the expected entry of emerging therapies.
DelveInsight’s “Head and Neck Cancer Market Insights, Epidemiology, and Market Forecast 2032” report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the Head and Neck Cancer market size, share, trends, and growth opportunities in the seven major markets (7MM) (i.e., the United States, EU4 (Germany, Spain, Italy, France), the United Kingdom and Japan).
The report covers emerging Head and Neck Cancer drugs, current treatment practices, market share of individual therapies, and current & forecasted market size from 2019 to 2032. It also evaluates the current Head and Neck Cancer treatment practice/algorithm, key drivers & barriers impacting the market growth, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the market.
Head and Neck Cancer: An Overview
Head and Neck Cancer (HNC) usually begins in the squamous cells – often referred to as squamous cell carcinomas of the head and neck – that line the mucosal surfaces of the head and neck (for example, those inside the mouth, throat, and voice box. They can also begin in the salivary glands, sinuses, muscles, or nerves, but they are less common than squamous cell carcinomas. HNC begins in several places in the head and throat, excluding brain or eye cancer.
The types of HNC include laryngeal cancer, nasopharyngeal cancer, hypopharyngeal cancer, nasal cavity and paranasal sinus cancer, salivary gland cancer, oral cancer, oropharyngeal cancer, and tonsil cancer. Histologically, progression to invasive HNSCC follows an ordered series beginning with epithelial cell hyperplasia, followed by dysplasia (mild, moderate, and severe), carcinoma in situ, and invasive carcinoma.
Symptoms of HNC include a lump in the neck or a sore in the mouth or throat that does not heal, a persistent sore throat, difficulty swallowing, and a change or hoarseness in the voice. Other, less dangerous illnesses can also cause these symptoms.
There are many tests used for diagnosing HNC- physical examination/blood and urine tests, endoscopy, biopsy, biomarker testing of the tumor, X-ray/barium swallow, panoramic radiograph, ultrasound, magnetic resonance imaging, bone scan, etc.
Head and Neck Cancer Market Key Facts
-
The Head and Neck Cancer market size in the 7MM was observed to be USD 2,801 million in 2021.
-
In 2021, the Head and Neck Cancer market size in the US was USD 1,831 million.
-
Among the EU-5 countries, Germany had the highest Head and Neck Cancer market size in 2021, i.e., USD 219 million. The lowest Head and Neck Cancer market size was estimated in Italy, with USD 114 million in 2021.
-
In Japan, the Head and Neck Cancer market size was observed to be USD 187 million in 2021.
-
In 2021, the total Head and Neck Cancer incident cases in the 7MM countries were ~158,000.
-
Among the European countries, Germany had the highest number of incident cases of Head and Neck Cancer in 2021, i.e., ~20,000 cases, followed by France which had ~16,000 incident cases in 2021. On the other hand, Italy had the lowest number of incident cases of Head and Neck Cancer, i.e. ~9900 cases in 2021. Japan had ~20,000 incident cases of HNC in 2021.

Head and Neck Cancer Market
The market outlook section of the report helps to build a detailed comprehension of the historical, current, and forecasted market size by analyzing the impact of current and emerging Head and Neck Cancer pipeline therapies. It also thoroughly assesses the Head and Neck Cancer market drivers & barriers, unmet needs, and emerging technologies set to impact the market dynamics.
The report gives complete details of the market trend for each marketed Head and Neck Cancer drug and mid & late-stage pipeline therapies by evaluating their impact based on the annual cost of therapy, their Mechanism of Action (MOA), Route of Administration (ROA), molecule types, competition with other therapies, brand value, and their impact on the market.
Head and Neck Cancer Epidemiology Assessment
The epidemiology section provides insights into the historical, current, and forecasted Head and Neck Cancer epidemiology trends in the seven major countries (7MM) from 2019 to 2032. It helps to recognize the causes of current and forecasted Head and Neck Cancer epidemiology trends by exploring numerous studies and research. The epidemiology section also provides a detailed analysis of diagnosed and prevalent patient pools, future trends, and views of key opinion leaders.
The Report Covers the Head and Neck Cancer Epidemiology, Segmented as –
-
Total Incident Cases of Head and Neck Cancer in the 7MM
-
Molecular Alteration-specific Incident Cases of Head and Neck Cancer in the 7MM
-
Stage-specific Incident Cases of Head and Neck Cancer in the 7MM
-
Site-specific Incident Cases of Head and Neck Cancer in the 7MM
-
HPV-specific Incident Cases of Head and Neck Cancer scenario in the 7MM
Head and Neck Cancer Drugs Uptake and Pipeline Development Activities
The drug uptake section focuses on the uptake rate of potential drugs recently launched in the Head and Neck Cancer market or expected to be launched during the study period. The analysis covers the Head and Neck Cancer market uptake by drugs, patient uptake by therapies, and sales of each drug. Moreover, the therapeutics assessment section helps understand the market dynamics by drug sales, the most rapid drug uptake, and the reasons behind the maximal use of particular drugs. Additionally, it compares the Head and Neck Cancer drugs based on their sale and market share.
The report also covers the Head and Neck Cancer pipeline development activities. It provides valuable insights about different therapeutic candidates in various stages and the key Head and Neck Cancer companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Learn How the Head and Neck Cancer Market Will Evolve and Grow by 2032 @
https://www.delveinsight.com/sample-request/head-and-neck-cancer-hnc-market
Head and Neck Cancer Therapeutics Analysis
Among all the available therapeutic options, chemotherapy is being extensively used in all lines of treatment. Multiple drugs have been identified with the advancement of medical technologies and increased research and development activities. Immunotherapeutic options have also been identified for treating HNC patients.
Overall, the treatment approach for Head and Neck Cancer (HNC) patients can involve multiple options like surgery, radiation therapy, chemotherapy, targeted therapy, and immunotherapy. Pharmacological therapies are mainly used to destroy cancer cells, and they are either administered systematically or locally (by a medical oncologist). Pharmacological therapies being administered to a HNC patient can be chemotherapy, targeted therapy, or immunotherapy.
In November 2011, the US FDA approved cetuximab (ERBITUX) in combination with chemotherapy for the first time for the treatment of Head and Neck Cancer (HNC) patients. The FDA approved pembrolizumab (KEYTRUDA) for the first-line treatment of patients with metastatic or unresectable recurrent HNSCC in June 2019. In April 2017, Bristol-Myers Squibb announced the approval of nivolumab (OPDIVO) as monotherapy for the treatment of SCCHN in adults.
Several major pharma and biotech companies are developing therapies for Head and Neck cancer. Currently, Innate Pharma is leading the therapeutics market with its Head and Neck cancer drug candidates in the most advanced stage of clinical development.
Head and Neck Cancer Companies Actively Working in the Therapeutics Market Include
Some of the key companies in the Head and Neck Cancer Companies Market include Debiopharm, Merck, AstraZeneca, MedImmune, Eisai Co Ltd/Merck & Co, Kura Oncology, Cellectar Biosciences, Galera Therapeutics, Inc., BioMimetix JV, LLC, Bristol-Myers Squibb, QBiotics Group Limited, Alentis Therapeutics AG, Coordination Pharmaceuticals, Inc., Genmab, Merck Sharp & Dohme LLC, Immunovative Therapies, Ltd., TyrNovo Ltd, BioNTech SE, Aveta Biomics, Inc., Pfizer, and Cue Biopharma, and other.
Emerging and Marketed Head and Neck Cancer Therapies Covered in the Report Include:
-
Durvalumab: AstraZeneca
-
Tipifarnib : Kura Oncology
-
Iopofosine: Cellectar Biosciences
-
TECENTRIQ (Atezolizumab): Hoffmann-La Roche
-
KEYTRUDA (pembrolizumab): Merck Sharp & Dohme
-
XEVINAPANT (debio 1143): Merck KGaA
-
Toripalimab: Junshi Biosciences/Coherus
-
Tipifarnib: Kura Oncology
And Many Others
Download the Sample Report to Learn More About the Key Companies and Emerging Therapies @
https://www.delveinsight.com/sample-request/head-and-neck-cancer-hnc-market
Table of Content (TOC)
1. Key Insights
2. Executive Summary
3. Head and Neck Cancer Competitive Intelligence Analysis
4. Head and Neck Cancer Market Overview at a Glance
5. Head and Neck Cancer Disease Background and Overview
6. Head and Neck Cancer Patient Journey
7. Head and Neck Cancer Patient Population and Epidemiology Trends (In the US, EU5, and Japan)
8. Head and Neck Cancer Treatment Algorithm, Current Treatment, and Medical Practices
9. Head and Neck Cancer Unmet Needs
10. Key Endpoints of Head and Neck Cancer Treatment
11. Head and Neck Cancer Marketed Therapies
12. Head and Neck Cancer Emerging Drugs and Latest Therapeutic Advances
13. Head and Neck Cancer Seven Major Market Analysis
14. Attribute Analysis
15. Head and Neck Cancer Market Outlook (In US, EU5, and Japan)
16. Head and Neck Cancer Companies Active in the Market
17. Head and Neck Cancer Access and Reimbursement Overview
18. KOL Views on the Head and Neck Cancer Market
19. Head and Neck Cancer Market Drivers
20. Head and Neck Cancer Market Barriers
21. Appendix
22. DelveInsight Capabilities
23. Disclaimer
*The Table of Contents (TOC) is not exhaustive; the final content may vary. Refer to the sample report for the complete table of contents.
Request the Sample PDF to Learn More About the Key Offerings of the Report @
https://www.delveinsight.com/sample-request/head-and-neck-cancer-hnc-market
Other Trending Healthcare Reports by DelveInsight
Epstein Barr virus (EBV) Market
“Epstein Barr virus (EBV) Market Insights, Epidemiology, and Market Forecast-2032” report delivers an in-depth understanding of the historical and forecasted epidemiology as well as the Epstein Barr virus (EBV) market size, share, trends, and growth analysis in the seven major markets (i.e. the United States, EU5 (Germany, Spain, Italy, France, and the United Kingdom) and Japan. Moreover, it also covers the current treatment practices, emerging drugs, market drivers, market barriers, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the Epstein Barr virus (EBV) market.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research Firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Shruti Thakur
Email: Send Email
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Head and Neck Cancer Market Poised for Phenomenal Expansion During the Forecast Period (2023-32) – Estimates DelveInsight | Debiopharm, Merck, AstraZeneca, MedImmune, Eisai, Merck, Kura Oncology





 The market outlook section of the report helps to build a detailed comprehension of the historical, current, and forecasted market size by analyzing the impact of current and emerging Angelman Syndrome pipeline therapies. It also thoroughly assesses the Angelman Syndrome market drivers & barriers, unmet needs, and emerging technologies set to impact the market dynamics.
The market outlook section of the report helps to build a detailed comprehension of the historical, current, and forecasted market size by analyzing the impact of current and emerging Angelman Syndrome pipeline therapies. It also thoroughly assesses the Angelman Syndrome market drivers & barriers, unmet needs, and emerging technologies set to impact the market dynamics.