The report covers emerging Peripheral Arterial Disease drugs, current treatment practices, market share of individual therapies, and current & forecasted market size from 2019 to 2032. It also evaluates the current Peripheral Arterial Disease treatment practice/algorithm, key drivers & barriers impacting the market growth, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the market.
Peripheral Arterial Disease: An Overview
Peripheral artery disease (PAD) is a common manifestation of arterial stenosis of the lower and/or upper extremities that reduces arterial flow. It is well known that Peripheral Arterial Disease is associated with elevated morbidity and mortality with cardiovascular (CV) disease.
In Peripheral Arterial Disease, the legs or arms—usually the legs—do not receive enough blood flow to keep up with the demand, which may cause leg pain when walking (claudication) and other symptoms. Many people with Peripheral Arterial Disease have no symptoms. The most common symptom of lower-extremity Peripheral Arterial Disease is painful muscle cramping in the hips, thighs, or calves when walking, climbing stairs, or exercising. However, those who develop a painful ache in their legs when they walk usually disappear after a few minutes of rest. The medical term for this is “intermittent claudication”.
Physical examination of the legs for symptoms like shiny skin, brittle toenails, hair loss on legs and feet, and leg ulcers are evaluated for the diagnosis of Peripheral Arterial Disease. The ankle-brachial pressure index (ABPI) test is widely used to diagnose Peripheral Arterial Disease. Further testing, like an ultrasound scan or an angiogram, is done when there is uncertainty about the diagnosis.
There is no cure for Peripheral Arterial Disease, but lifestyle changes and medicine can help reduce the symptoms. Medications called statins are commonly prescribed. If Peripheral Arterial Disease is so far progressed and does not respond to noninvasive therapies such as lifestyle changes, medication, or both, surgery may be necessary.
Peripheral Arterial Disease Market Key Facts
-
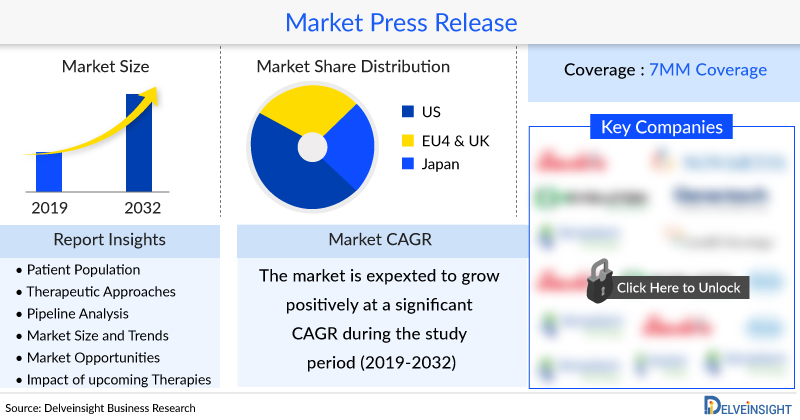
The total market size of Peripheral Arterial Disease in the US was approximately USD 2559 million in 2022.
-
In 2022, EU4 and the UK countries captured an approximately estimated USD 989 million.
-
Among EU4 and the UK, Germany consistently captured the highest market, with approximately USD 397 million in 2022.
-
Among the 7MM, Japan represented approximately 10% of the total market of Peripheral Arterial Disease with USD 382 million in 2022.
-
In 7MM, there were approximately 17,245,750, diagnosed prevalent cases of Peripheral Arterial Disease in 2022, which are expected to increase at a significant CAGR by 2032.
-
The US accounted for approximately 9,505,662 cases, which was the highest diagnosed prevalent cases of Peripheral Arterial Disease, followed by EU4 and the UK with 5,906,659 cases, and Japan with 1,833,429 cases in 2022. These cases are expected to increase in the US, EU4, the UK, and Japan by 2032.
-
Among the European countries, Germany had the highest diagnosed prevalent population of Peripheral Arterial Disease (approximately 2,371,290 cases), followed by Italy (1,023,681 cases) in 2022. On the other hand, Spain (767,393 cases) had the lowest diagnosed prevalent population in EU4 and the UK countries and the 7MM.

Peripheral Arterial Disease Market
The market outlook section of the report helps to build a detailed comprehension of the historical, current, and forecasted market size by analyzing the impact of current and emerging Peripheral Arterial Disease pipeline therapies. It also thoroughly assesses the Peripheral Arterial Disease market drivers & barriers, unmet needs, and emerging technologies set to impact the market dynamics.
The report gives complete details of the market trend for each marketed Peripheral Arterial Disease drug and mid & late-stage pipeline therapies by evaluating their impact based on the annual cost of therapy, their Mechanism of Action (MOA), Route of Administration (ROA), molecule types, competition with other therapies, brand value, and their impact on the market.
Peripheral Arterial Disease Epidemiology Assessment
The epidemiology section provides insights into the historical, current, and forecasted Peripheral Arterial Disease epidemiology trends in the seven major countries (7MM) from 2019 to 2032. It helps to recognize the causes of current and forecasted Peripheral Arterial Disease epidemiology trends by exploring numerous studies and research. The epidemiology section also provides a detailed analysis of diagnosed and prevalent patient pools, future trends, and views of key opinion leaders.
The Report Covers the Peripheral Arterial Disease Epidemiology, Segmented as –
-
Total Diagnosed Prevalent Cases of Peripheral Arterial Disease in the 7MM [2019–2032]
-
Age-specific Diagnosed Prevalent Cases of Peripheral Arterial Disease in the 7MM [2019–2032]
-
Severity-specific Diagnosed Prevalent Cases of Peripheral Arterial Disease in the 7MM [2019–2032]
-
Gender-specific Diagnosed Prevalent Cases of Peripheral Arterial Disease in the 7MM [2019–2032]
Peripheral Arterial Disease Drugs Uptake and Pipeline Development Activities
The drug uptake section focuses on the uptake rate of potential drugs recently launched in the Peripheral Arterial Disease market or expected to be launched during the study period. The analysis covers the Peripheral Arterial Disease market uptake by drugs, patient uptake by therapies, and sales of each drug. Moreover, the therapeutics assessment section helps understand the market dynamics by drug sales, the most rapid drug uptake, and the reasons behind the maximal use of particular drugs. Additionally, it compares the Peripheral Arterial Disease drugs based on their sale and market share.
The report also covers the Peripheral Arterial Disease pipeline development activities. It provides valuable insights about different therapeutic candidates in various stages and the key Peripheral Arterial Disease companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Learn How the Peripheral Arterial Disease Market Will Evolve and Grow by 2032 @
Peripheral Arterial Disease Therapeutics Analysis
Several major pharma and biotech companies are developing therapies for Peripheral Arterial Disease. Currently, Novo Nordisk is leading the therapeutics market with its Peripheral Arterial Disease drug candidates in the most advanced stage of clinical development.
Peripheral Arterial Disease Companies Actively Working in the Therapeutics Market Include
-
Novo Nordisk
-
Takeda
-
Mercator MedSystems, Inc.
-
Beijing Northland Biotech. Co., Ltd.
-
Ixaka Ltd
-
Humacyte, Inc.
-
CardioVascular BioTherapeutics
-
Proteon Therapeutics
-
ReNeuron Limited
-
Alucent Biomedical
-
Athersys
-
ARCA biopharma
-
Ambulero
-
Venturis Therapeutics
And Many Others
Emerging and Marketed Peripheral Arterial Disease Therapies Covered in the Report Include:
-
Semaglutide: Novo Nordisk
-
VM202: Helixmith
And Many More
Download the Sample Report to Learn More About the Key Companies and Emerging Therapies @
Table of Content (TOC)
1. Key Insights
2. Executive Summary
3. Peripheral Arterial Disease Competitive Intelligence Analysis
4. Peripheral Arterial Disease Market Overview at a Glance
5. Peripheral Arterial Disease Disease Background and Overview
6. Peripheral Arterial Disease Patient Journey
7. Peripheral Arterial Disease Patient Population and Epidemiology Trends (In the US, EU5, and Japan)
8. Peripheral Arterial Disease Treatment Algorithm, Current Treatment, and Medical Practices
9. Peripheral Arterial Disease Unmet Needs
10. Key Endpoints of Peripheral Arterial Disease Treatment
11. Peripheral Arterial Disease Marketed Therapies
12. Peripheral Arterial Disease Emerging Drugs and Latest Therapeutic Advances
13. Peripheral Arterial Disease Seven Major Market Analysis
14. Attribute Analysis
15. Peripheral Arterial Disease Market Outlook (In US, EU5, and Japan)
16. Peripheral Arterial Disease Companies Active in the Market
17. Peripheral Arterial Disease Access and Reimbursement Overview
18. KOL Views on the Peripheral Arterial Disease Market
19. Peripheral Arterial Disease Market Drivers
20. Peripheral Arterial Disease Market Barriers
21. Appendix
22. DelveInsight Capabilities
23. Disclaimer
*The Table of Contents (TOC) is not exhaustive; the final content may vary. Refer to the sample report for the complete table of contents.
Request the Sample PDF to Learn More About the Key Offerings of the Report @
Other Trending Healthcare Reports by DelveInsight
Dermal Regeneration Matrix Market
“Dermal Regeneration Matrix Market Insights, Epidemiology, and Market Forecast-2032” report delivers an in-depth understanding of the historical and forecasted epidemiology as well as the Dermal Regeneration Matrix market size, share, trends, and growth analysis in the seven major markets (i.e. the United States, EU5 (Germany, Spain, Italy, France, and the United Kingdom) and Japan. Moreover, it also covers the current treatment practices, emerging drugs, market drivers, market barriers, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the Dermal Regeneration Matrix market.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research Firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Shruti Thakur
Email: Send Email
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Peripheral Arterial Disease Market: Breakthrough Forecast Study Predicts Astounding Growth in the Upcoming Years | Novo Nordisk, Takeda, Ixaka, Humacyte, Proteon Therapeutics, ReNeuron Limited


 The market outlook section of the report helps to build a detailed comprehension of the historical, current, and forecasted market size by analyzing the impact of current and emerging Angelman Syndrome pipeline therapies. It also thoroughly assesses the Angelman Syndrome market drivers & barriers, unmet needs, and emerging technologies set to impact the market dynamics.
The market outlook section of the report helps to build a detailed comprehension of the historical, current, and forecasted market size by analyzing the impact of current and emerging Angelman Syndrome pipeline therapies. It also thoroughly assesses the Angelman Syndrome market drivers & barriers, unmet needs, and emerging technologies set to impact the market dynamics.