The extensive report offers a thorough examination of the market potential and market share of PALYNZIQ (Pegvaliase-pqpz) in the therapeutics landscape for Phenylketonuria across the 7MM, spanning the years 2019 to 2032. By delving into this analysis, stakeholders gain valuable insights into the evolving landscape of PALYNZIQ (Pegvaliase-pqpz), encompassing both clinical and commercial dimensions. Key parameters such as the drug’s Mechanism of Action (MOA), Route of Administration (ROA), dosage, and any special designations are meticulously explored, providing a holistic understanding of its profile.
PALYNZIQ (Pegvaliase-pqpz) Drug Insights
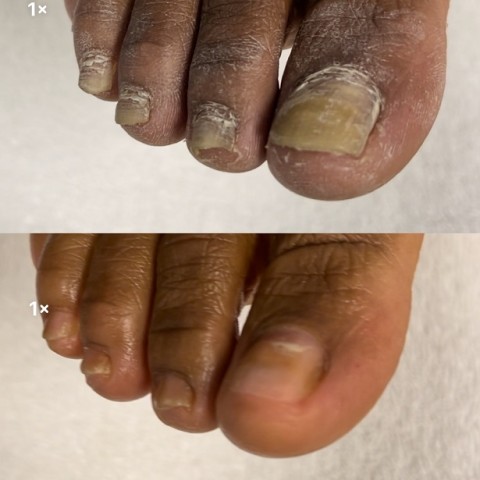
PALYNZIQ (Pegvaliase-pqpz) Injection marks a groundbreaking milestone as the inaugural FDA-endorsed enzyme substitution therapy tailored for adult individuals grappling with phenylketonuria (PKU), wherein their blood phenylalanine (Phe) levels soar beyond 600 µmol/L (10 mg/dL) despite ongoing treatment. This novel therapy, administered once daily through self-administration, operates autonomously of the Phenylalanine Hydroxylase (PAH) enzyme, rendering it a viable treatment avenue for all qualifying adult patients contending with PKU. This approval heralds a significant advancement in the management of PKU, offering newfound hope and enhanced therapeutic options to individuals grappling with this metabolic disorder.
Mechanism of Action of PALYNZIQ (Pegvaliase-pqpz)
Pegvaliase-pqpz stands as a PEGylated phenylalanine ammonia-lyase (PAL) enzyme meticulously designed to catalyze the conversion of phenylalanine into ammonia and trans-cinnamic acid. This innovative enzyme serves as a crucial substitute for the impaired activity of the Phenylalanine Hydroxylase (PAH) enzyme in individuals grappling with phenylketonuria (PKU), thereby facilitating a reduction in blood phenylalanine (Phe) concentrations. By effectively stepping in to perform the function of the deficient PAH enzyme, pegvaliase-pqpz presents a promising therapeutic avenue for managing PKU and potentially mitigating associated health complications. This targeted enzymatic intervention represents a significant stride in the quest for improved treatments and outcomes for individuals living with PKU.
Explore key clinical, commercial, and regulatory milestones associated with PALYNZIQ (Pegvaliase-pqpz) by visiting:
Key Highlights of the PALYNZIQ (Pegvaliase-pqpz) Market Report
- The report includes a projected assessment of PALYNZIQ (Pegvaliase-pqpz) sales for Phenylketonuria up to the year 2032.
- The report offers in-depth coverage of advanced-stage emerging therapies for the treatment of Phenylketonuria.
- The report additionally includes both qualitative and quantitative analyses conducted by analysts, along with Key Opinion Leader (KOL) perspectives on PALYNZIQ (Pegvaliase-pqpz) for Phenylketonuria.
Why PALYNZIQ (Pegvaliase-pqpz) Market Report?
- The projected market data for PALYNZIQ (Pegvaliase-pqpz) in the context of Phenylketonuria will play a crucial role in informing decision-making related to the therapeutic portfolio. It will provide valuable insights into the overall landscape of PALYNZIQ (Pegvaliase-pqpz), aiding in strategic planning and decision-making processes within the therapeutic domain.
- A comprehensive market forecast for PALYNZIQ (Pegvaliase-pqpz) will contribute to a better understanding of how the drug competes with other emerging therapies within the therapeutics landscape. This analysis will provide insights into the drug’s positioning, potential market share, and overall impact on the competitive environment in the therapeutic field.
- The report additionally offers future market assessments for the PALYNZIQ (Pegvaliase-pqpz) market in the field of Phenylketonuria across the 7 Major Markets. It encompasses advanced qualitative analyses such as SWOT analysis, insights from expert analysts, a comprehensive overview of market competitors, and a brief analysis of other emerging therapies within the realm of Phenylketonuria. This multifaceted approach ensures a comprehensive understanding of the PALYNZIQ (Pegvaliase-pqpz) market landscape, combining both quantitative and qualitative perspectives for a well-rounded assessment.
- Conducting a thorough market forecast for PALYNZIQ (Pegvaliase-pqpz) will facilitate a detailed analysis of the drug’s clinical trial progress. This examination will encompass a comprehensive assessment of the clinical, regulatory, and commercial aspects, providing valuable insights into the advancements of PALYNZIQ (Pegvaliase-pqpz).
Visit and Explore How PALYNZIQ (Pegvaliase-pqpz) Is Set to Dominate the Phenylketonuria Therapeutic Market:
Table of Contents of the Report
1. Report Introduction
2. PALYNZIQ (Pegvaliase-pqpz) Overview in Phenylketonuria
3. Competitive Landscape (Key Assessment of the Marketed Therapies)
4. Competitive Landscape (Late-stage Emerging Therapies Analysis)
5. PALYNZIQ (Pegvaliase-pqpz) Market Assessment
6. SWOT Analysis
7. Analysts’ Views
8. Appendix
9. DelveInsight Capabilities
10. Disclaimer
11. About DelveInsight
* The final table of contents may be subject to change based on user demand.
Request the Sample PDF to Learn More About the Key Offerings of the PALYNZIQ (Pegvaliase-pqpz) Market Report @
Other Related Reports By DelveInsight
Phenylketonuria Pipeline Insight
DelveInsight’s “Phenylketonuria Pipeline Insight” report provides comprehensive insights about 18+ companies and 20+ pipeline drugs in the Phenylketonuria pipeline landscape. It covers the pipeline drug profiles, including clinical and nonclinical stage products by routes of administration, and molecule types. Furthermore, it further highlights the inactive pipeline products in this space. Some of the key companies in the Phenylketonuria Therapeutics market include BioMarin Pharmaceutical, Homology Medicine, Jnana Therapeutics, Nestlé Health Science, Moderna, SOM Biotech, Agios Pharmaceuticals, APR Applied Pharma Research, American Gene Technologies, Generation Bio, and PTC Therapeutics, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Shruti Thakur
Email: Send Email
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/oncology
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: PALYNZIQ (Pegvaliase-pqpz) Market Size and Share Analysis Across 7MM and Competitive Landscape by DelveInsight | Key Players – BioMarin Pharmaceutical