It’s common for footwear to deteriorate, especially in the area of the insoles, due to factors like sweat, moisture, and inadequate drying. When shoes or boots get wet, whether from sweat or external sources, and aren’t thoroughly dried, it creates an environment conducive to bacterial and fungal growth. Over time, this can lead to the breakdown of materials, including the insoles, causing them to deteriorate and potentially rot.
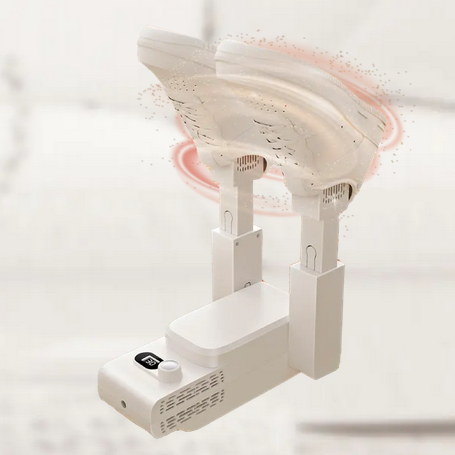
Qinux DizaDry emerges as a specialized electric shoe dryer meticulously crafted to provide comprehensive ventilation to footwear from within, utilizing hot air to ensure thorough and efficient drying. Tailored for periods of inclement weather when rain and snow render footwear damp, this innovative device offers an ideal solution to combat moisture and maintain shoe integrity.
Designed with precision, Qinux DizaDry boasts a user-friendly interface, allowing individuals to effortlessly dry their shoes with optimal efficiency. By circulating hot air throughout the interior of the footwear, it effectively evaporates moisture, leaving shoes dry and ready for use in a fraction of the time.
This electric shoe dryer is particularly beneficial for individuals living in regions prone to frequent precipitation or those engaging in outdoor activities where shoes are susceptible to becoming wet. Whether it’s hiking boots soaked from traversing muddy trails or sneakers dampened by unexpected rainfall, Qinux DizaDry offers a convenient and reliable method to restore footwear to its pristine condition.
Moreover, its compact and portable design ensures versatility and ease of use, making it suitable for home use or travel purposes. With Qinux DizaDry at their disposal, users can bid farewell to the inconvenience of damp footwear and confidently step out into any weather condition, knowing their shoes will remain dry and comfortable throughout the day.
By following the simple steps bellow, Qinux DizaDry provides a convenient and efficient solution for drying footwear, ensuring that shoes or boots remain dry and comfortable, especially during periods of wet weather.
- Step 1: Insert Qinux DizaDry into the footwear that needs to be dried, ensuring it fits securely within the shoes or boots.
- Step 2: Power On: Plug the device into a power source and activate it using the power switch or button located on the device.
- Step 3: Hot Air Circulation: Qinux DizaDry generates hot air, which is then circulated throughout the interior of the footwear.
- Step 4: Ventilation: The hot air is evenly distributed inside the shoes or boots, reaching all areas including the toe box, insoles, and uppers.
- Step 5: Moisture Evaporation: The circulating hot air effectively evaporates any moisture present within the footwear, including sweat, rainwater, or snowmelt.
- Step 6: Drying Process: Over time, the continuous circulation of hot air gradually dries the footwear, removing all moisture.
- Step 7: Completion: Once the drying process is complete, power off the device and remove it from the footwear.
- Step 8: Ready for Use: The footwear is now dry and ready for immediate use, ensuring comfort and protection for the wearer.
Qinux DizaDry offers a range of features designed to provide efficient, convenient, and reliable drying for footwear, ensuring that shoes and boots remain dry, comfortable, and ready for use in any weather condition. Here are the features of Qinux DizaDry:
Efficient Drying: Utilizes hot air circulation to efficiently dry footwear from the inside out, eliminating moisture and reducing the risk of odors and mold.
Compact and Portable: Designed to be compact and lightweight, making it easy to carry and store, ideal for home use or travel.
Versatile Compatibility: Fits into a wide range of footwear sizes and styles, accommodating various types of shoes, boots, and athletic footwear.
User-Friendly Operation: Simple power switch or button for easy activation and deactivation, with intuitive controls for hassle-free use.
Safe and Reliable: Built-in safety features to prevent overheating or damage to footwear, ensuring safe operation.
Energy-Efficient: Designed to use minimal power while providing effective drying, reducing energy consumption and saving on electricity costs.
Durable Construction: Made from high-quality materials for long-lasting durability and reliability, ensuring years of use.
Multipurpose Usage: Can also be used to dry gloves, socks, and other small garments or accessories, offering versatility in drying options.
Quiet Operation: Operates quietly, allowing for discreet use without disturbing others nearby.
Effortless Maintenance: Easy to clean and maintain, with removable components for convenient upkeep and longevity of the device.
- Effectively eliminates moisture from footwear, reducing drying time and preventing discomfort.
- Helps prevent the formation of unpleasant odors by thoroughly drying shoes and boots after wear.
- Reduces the risk of mold and mildew growth by removing excess moisture from footwear.
- Prolongs the lifespan of shoes and boots by preventing deterioration caused by moisture damage.
- Compact and lightweight design allows for easy transport and use at home or while traveling.
- Can be used with various types of footwear, including shoes, boots, and athletic shoes, for versatile drying options.
- Operates with minimal power consumption, helping to save on electricity costs while providing effective drying.
- Ensures dry and comfortable footwear, promoting foot health and overall comfort throughout the day,
- Operates quietly, allowing for discreet use without causing disturbance to others nearby.
- Easy to clean and maintain, with removable components for effortless upkeep and long-term durability.
At $89, Qinux DizaDry offers an affordable yet effective solution for maintaining dry and comfortable footwear. With this investment, individuals can elevate their shoe care routine, bidding farewell to the inconvenience of damp, uncomfortable shoes. The product is available in stock at its official website.
The device’s ease of use, quiet operation, and effective drying capabilities make it a highly recommended purchase for those seeking convenient and reliable footwear maintenance solutions. It accommodates any boot or shoe with ease, providing a snug fit for all footwear types. Moreover it helps maintain shoe and indoor hygiene by eliminating bacteria and bad smell from the shoes. Notably, its silent operation guarantees minimal disruption, allowing users to enjoy dry and warm shoes without disturbance. Overall, based on user experience, many find it to be a worthwhile investment, all glory to its efficient performance and user-friendly design.
Media Contact
Company Name: Qinux DizaDry
Contact Person: Media Relations
Email: Send Email
Country: United States
Website: https://rebrand.ly/DizaDry
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Qinux DizaDry Launches Portable Shoe Dryer Sterilizer