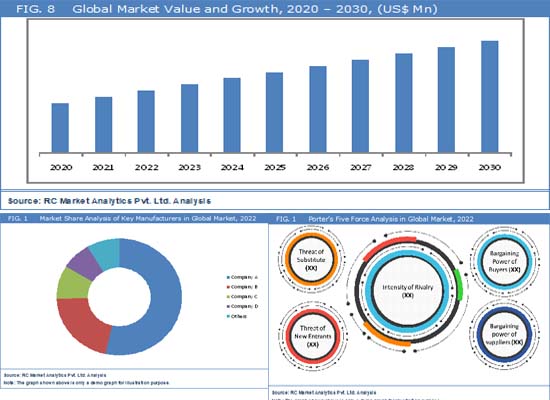
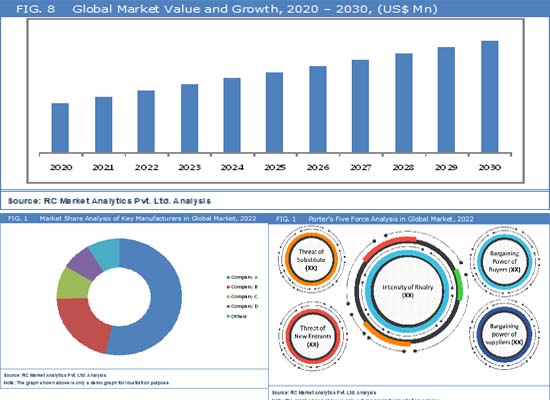
The projected period from 2024 to 2030 foresees a substantial growth, with an estimated noteworthy Compound Annual Growth Rate (CAGR) of approximately 6% in the global automotive traction control systems market. Stringent safety regulations on a global scale are compelling automakers to integrate traction control systems, ensuring both vehicle stability and compliance with safety standards. The continual expansion of global vehicle production, especially in emerging markets, is driving the demand for these systems as standard features in contemporary vehicles. Consumer preferences for advanced safety features are contributing to the increasing adoption of traction control systems, enhancing vehicle stability during acceleration, braking, and cornering. Ongoing technological advancements in sensors and electronic control units (ECUs) are further improving the reliability and performance of these systems.
Market Trends:
- Rising demand for safety features: Consumers are increasingly prioritizing safety features like traction control when making car purchase decisions, creating a favorable market environment for these systems.
- Focus on cost reduction: Manufacturers are investing in research and development to optimize production processes and reduce the cost of traction control systems, making them more affordable for a wider range of vehicles.
- Growing competition: The market is witnessing increasing competition among established players and new entrants, leading to innovation and a wider variety of traction control systems available.
- Integration with telematics: Connectivity features are being incorporated into traction control systems, allowing for remote monitoring and data analysis for improved performance and predictive maintenance.
- Increased demand in emerging markets: Growing economies in regions like Asia Pacific and Latin America are expected to see a significant rise in demand for traction control systems due to rising disposable incomes and increased focus on vehicle safety.
According to the recent report published by RC Market Analytics, the Global Automotive Traction Control Systems Market is expected to provide sustainable growth opportunities during the forecast period from 2023 to 2030. This latest industry research study analyzes the automotive traction control systems market by various product segments, applications, regions and countries while assessing regional performances of numerous leading market participants. The report offers a holistic view of the automotive traction control systems industry encompassing numerous stakeholders including raw material suppliers, providers, distributors, consumers and government agencies, among others. Furthermore, the report includes detailed quantitative and qualitative analysis of the global market considering market history, product development, regional dynamics, competitive landscape, and key success factors (KSFs) in the industry.
Browse the Full Report Discretion @ https://www.rcmarketanalytics.com/automotive-traction-control-systems-market/
Geographically, the automotive traction control systems market report comprises dedicated sections centering on the regional market revenue and trends. The automotive traction control systems market has been segmented on the basis of geographic regions into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. Automotive traction control systems market estimates have also been provided for the historical years 2020 & 2021 along with forecast for the period from 2023 – 2030.The report includes a deep-dive analysis of key countries including the U.S., Canada, the U.K., Germany, France, Italy, China, Japan, India, Australia, Mexico, Brazil and South Africa, among others. Thereby, the report identifies unique growth opportunities across the world based on trends occurring in various developed and developing economies.
The Automotive Traction Control Systems Market Segmentation:
By Type:
- Mechanical System
- Electric System
By Component:
- Hydraulic Modulators
- ECU
- Sensors
- Others
By Vehicle Type:
- Passenger Vehicle
- Commercial Vehicle
By Sales Channel:
- OEM
- Aftermarket
By Region:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Key players in the worldwide automotive traction control systems market include Bosch GmbH, Continental AG, ZF Friedrichshafen AG, Denso Corporation, and Aisin Seiki Co., Ltd. These companies are adopting strategies such as expansion, new investments, the introduction of innovative services, and collaboration to navigate markets effectively. In their pursuit of a competitive edge, players are actively expanding into new geographical areas through both organic growth and strategic acquisitions, leveraging joint synergies to strengthen their market positions.
To know more about this study, request a free sample report @ https://www.rcmarketanalytics.com/request-sample/?id=857
Key Questions Answered by Automotive Traction Control Systems Market Report:
- Product popularity and adoption based on various country-level dynamics
- Regional presence and product development for leading market participants
- Market forecasts and trend analysis based on ongoing investments and economic growth in key countries
- Competitive landscape based on revenue, product offerings, years of presence, number of employees and market concentration, among others
- Various industry models such as SWOT analysis, Pestle Analysis, Porter’s Five Force model, Value Chain Analysis pertaining to Automotive Traction Control Systems market
- Analysis of the key factors driving and restraining the growth of the global, regional and country-level markets from 2020-2030
About Us: RC Market Analytics is a global market research firm. Our insightful analysis is focused on developed and emerging markets. We identify trends and forecast markets with a view to aid businesses identify market opportunities to optimize strategies. Our expert’s team of analysts’ provides enterprises with strategic insights. RC Market Analytics works to help enterprises grow through strategic insights and actionable solutions. Feel free to contact us for any report customization at sales@researchcorridor.com.
Media Contact
Company Name: RC Market Analytics Pvt. Ltd.
Contact Person: Vijendra Singh
Email: Send Email
Country: India
Website: https://www.researchcorridor.com/
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: Automotive Traction Control Systems Market Size, Growth & Statistics Report to 2030